Home >
News > Ultrathin Mesoporous Metal–Organic Framework Nanosheets
Ultrathin Mesoporous Metal–Organic Framework Nanosheets
This study reports a novel bottom‑up interface‑directed co‑assembly strategy for synthesizing ultrathin two‑dimensional mesoporous UiO‑66(Ce) metal–organic framework nanosheets. The method employs amphiphilic perfluorooctanoic acid (PFCA) lipid bilayers and PS‑b‑PEO block copolymer spherical micelles as dual soft templates to form sandwich‑like 2D assemblies, which guide the confined growth of MOFs. The resultant 2D‑mUiO‑66(Ce) features uniform ~23 nm mesopores and a tunable thickness from 3 to 150 nm. Benefiting from through‑mesopore channels, shortened mass‑transport paths, abundant exposed Ce³⁺/Ce⁴⁺ active sites, and a narrowed band gap, the material exhibits significantly enhanced photocatalytic U(VI) reduction efficiency compared with bulk UiO‑66(Ce) and mesoporous mUiO‑66(Ce) particles. This work provides a new route to fabricate continuous 2D mesoporous MOFs and highlights the morphology and mesostructure effects in boosting photocatalytic environmental remediation.
Research Background
1. Key Problems in the Field
Traditional bulk MOFs are mostly microporous, leading to slow mass transport, limited molecular accessibility, and low utilization of active sites, especially for large guest molecules.
Most reported mesoporous MOFs are spherical or bulk solids rather than well‑defined 2D nanosheets; existing 2D mesoporous MOFs are usually aggregates of nanoparticles with slit‑shaped, poorly interconnected pores and thickness above 30 nm.
Continuous, ultrathin 2D mesoporous MOFs with through‑pores and controllable thickness have not been successfully synthesized, restricting their performance in adsorption, catalysis, and environmental remediation.
Cerium‑based MOFs possess unique redox properties and stability, but their morphological and porous engineering remains insufficient for high‑efficiency photocatalysis.
2. Previous Solutions by Other Researchers
Bottom‑up self‑assembly methods using block copolymers or surfactants as templates have produced various mesoporous MOFs with improved pore connectivity.
2D MOF nanosheets have been fabricated via exfoliation, interface growth, and anisotropic synthesis, showing enhanced electron transport and surface exposure.
Ce‑based MOFs have been explored in photocatalysis, redox reactions, and pollutant removal owing to Ce³⁺/Ce⁴⁺ mixed valence and oxygen storage capacity.
3. Novel Ideas and Innovations of This Work
Develop a dual‑template interface co‑assembly method using PFCA bilayers and PS‑b‑PEO micelles to construct continuous ultrathin 2D mesoporous MOF nanosheets for the first time.
Realize precise control over sheet thickness (3–150 nm) and uniform ~23 nm through‑mesopores.
Integrate 2D morphology, mesoporosity, and Ce redox sites to boost photocatalytic U(VI) reduction via improved mass transport and charge separation.
Combine experimental characterization with DFT and MD simulations to reveal band‑gap modulation and ion‑transport mechanisms.
Experimental Content
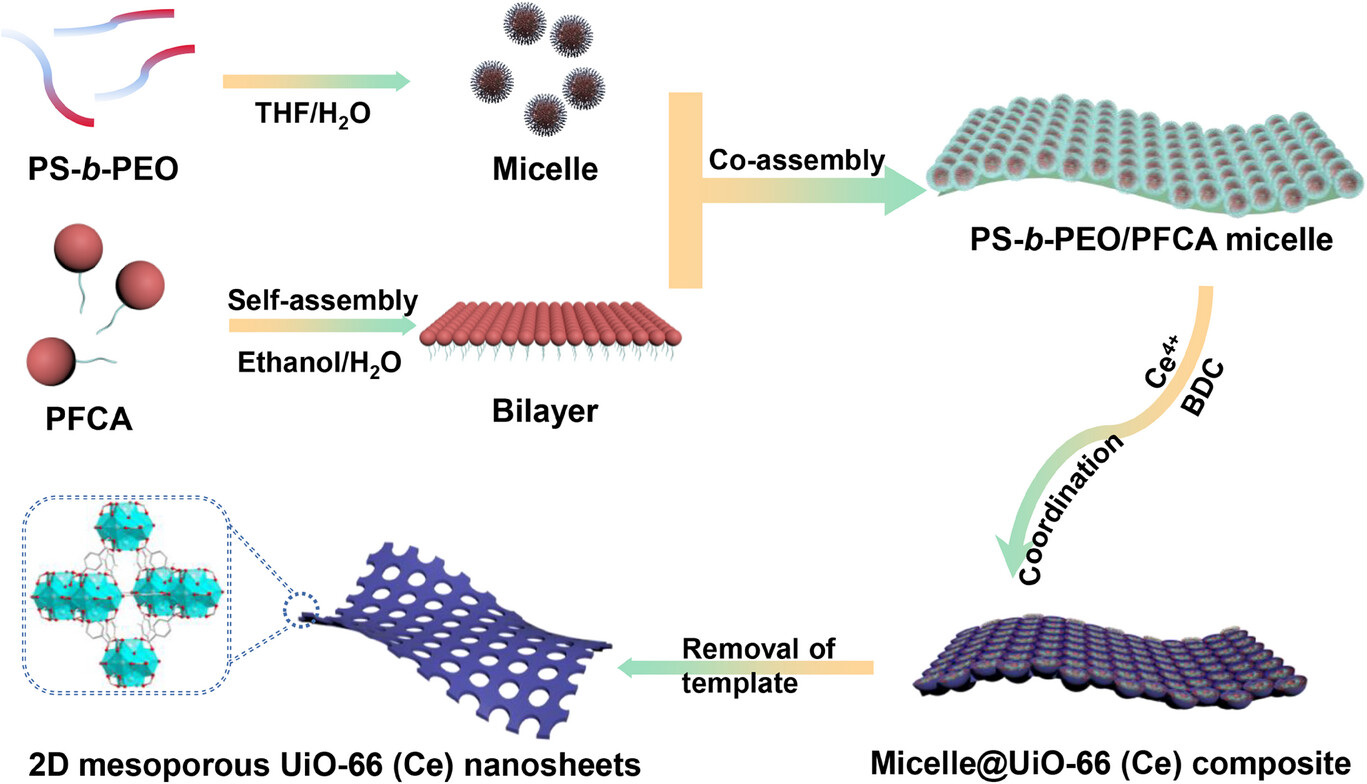
1. Synthesis of 2D‑mUiO‑66(Ce) Nanosheets
PS₉₅₀₀‑b‑PEO₅₀₀₀ was dissolved in THF, followed by gradual water addition to form micelles.
PFCA ethanol solution was introduced to form 2D bilayers; acetic acid and NaClO₄ were added as coordination modulators.
(NH₄)₂Ce(NO₃)₆ and terephthalic acid (BDC) were added, and the mixture reacted at 60 °C for 15 min.
The product was centrifuged, washed, and vacuum‑dried to obtain 2D‑mUiO‑66(Ce). Thickness was tuned by varying BDC dosage (33 mg for ~3 nm, 66 mg for ~13 nm, 198 mg for ~150 nm).
2. Preparation of Reference Samples
mUiO‑66(Ce): Synthesized under the same conditions without PFCA, forming cubic mesoporous particles.
UiO‑66(Ce): Prepared via a conventional solvothermal method without templates, yielding nonporous microporous cubic particles.
3. Photocatalytic U(VI) Reduction Tests
Reaction system: 5 mg catalyst, 45 mL of 50 ppm U(VI) solution, 5 mL methanol as sacrificial agent, 300 W xenon lamp irradiation.
Dark adsorption for 150 min followed by light irradiation for 200 min; U(VI) concentration was determined by Arsenazo III spectrophotometry at 652 nm.
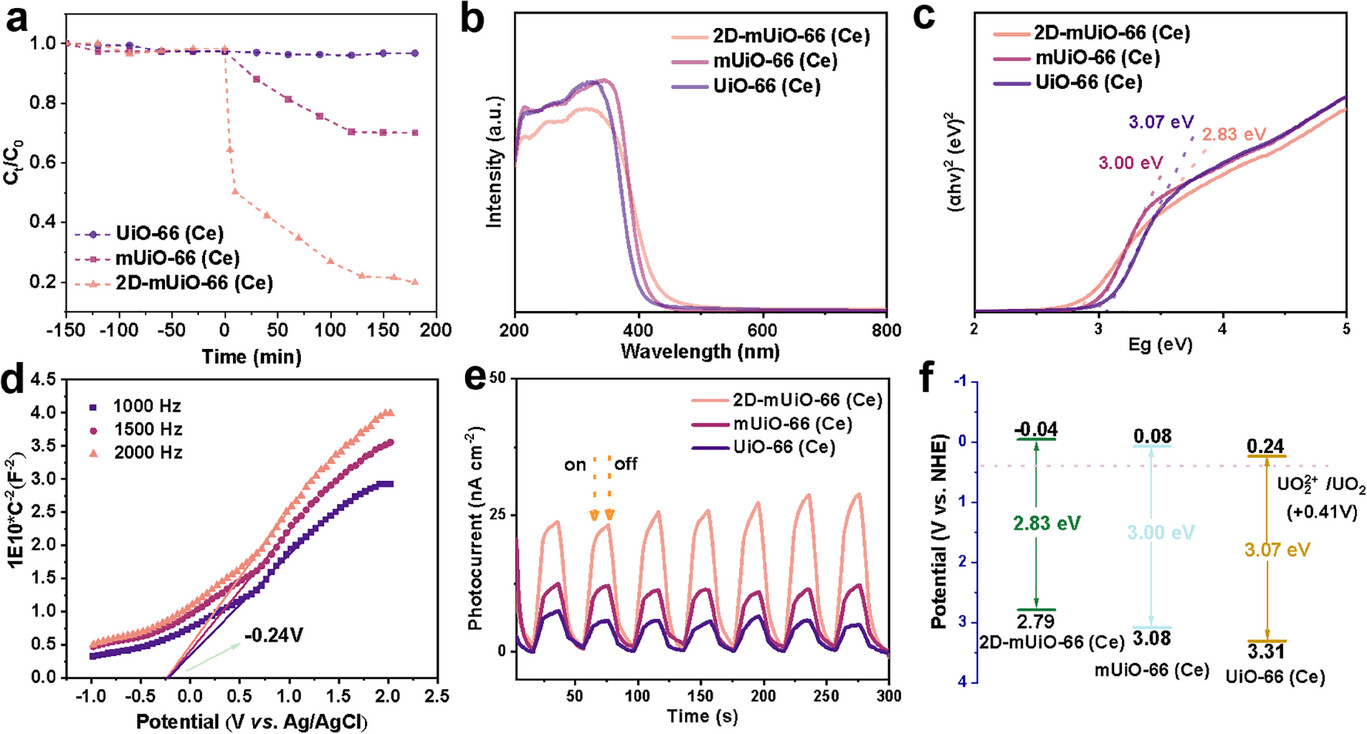
Key result: 2D‑mUiO‑66(Ce) removed 80.3% U(VI) within 200 min, far exceeding mUiO‑66(Ce) (30.0%) and UiO‑66(Ce).
4. Stability and Recyclability
After photocatalysis, the catalyst maintained sheet morphology and crystallinity without structural collapse, confirming good durability.
5. Theoretical Simulations
DFT calculations on 5 models (3D‑UiO‑66(Ce), 3D‑spUiO‑66(Ce), 2D‑UiO‑66(Ce), 2D‑spUiO‑66(Ce), 2D‑lpUiO‑66(Ce)) to analyze band‑gap evolution.
MD simulations to evaluate Na⁺/Cl⁻ ion transport through 2D frameworks with different pore sizes.
Characterization and Analysis
1. Morphology and Microstructure
SEM/TEM: 2D‑mUiO‑66(Ce) shows micrometer‑scale sheet morphology with uniform ~23 nm through‑mesopores; AFM confirms thickness of ~13 nm (standard sample).
Elemental mapping: Ce, C, O distribute homogeneously across the nanosheet.
Control experiments: Without PFCA, cubic mUiO‑66(Ce) particles form; without PS‑b‑PEO, no MOF product is obtained.
2. Crystal Structure
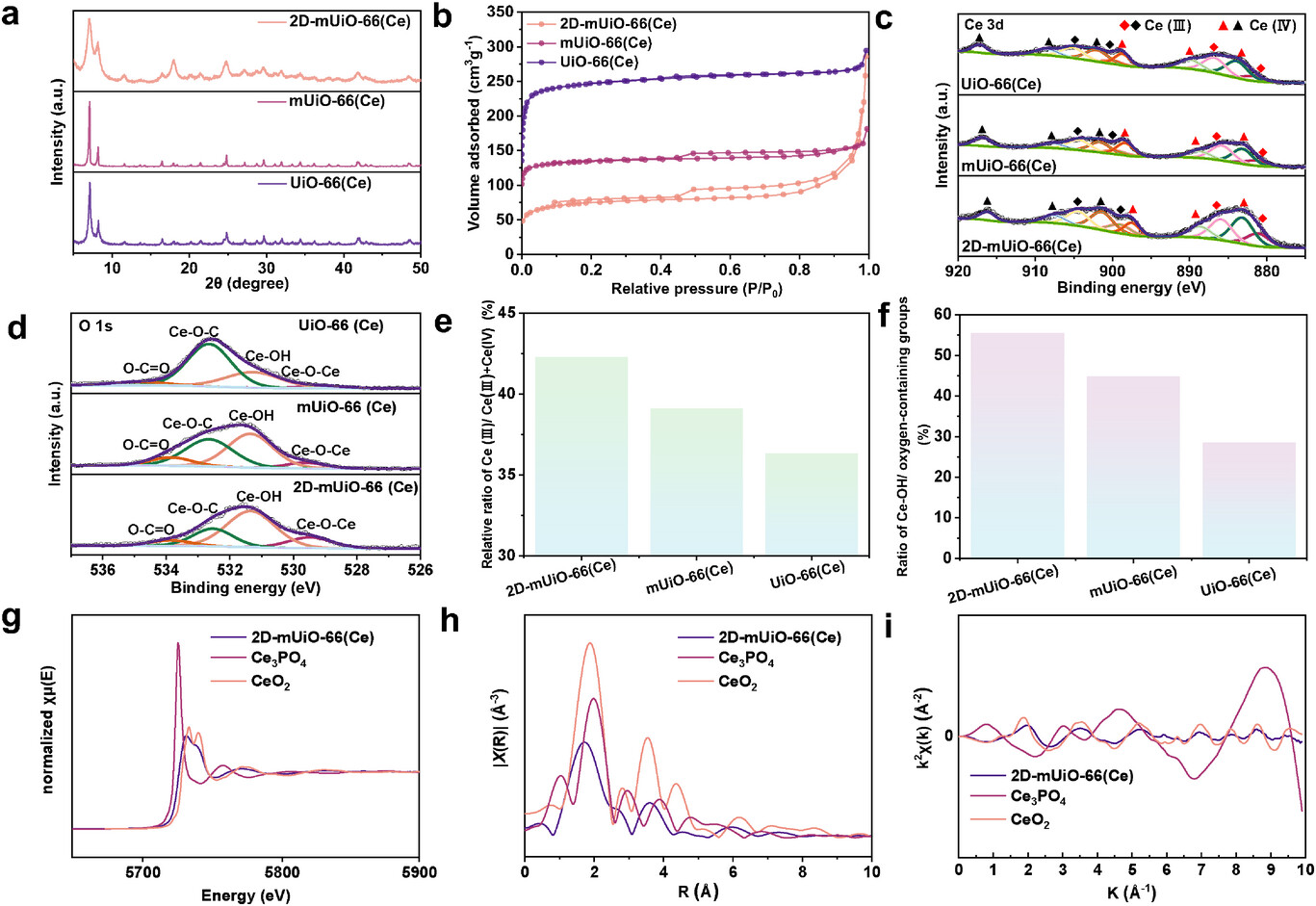
XRD: 2D‑mUiO‑66(Ce) matches the face‑centered cubic topology of UiO‑66(Ce), but peaks are broadened with reduced intensity due to confined growth and structural defects.
3. Porous Properties
N₂ adsorption–desorption: Type IV isotherm with obvious hysteresis, confirming mesoporosity.
BET surface area: 278.9 m² g⁻¹; total pore volume: 0.403 cm³ g⁻¹.
Comparison: mUiO‑66(Ce) 503.32 m² g⁻¹; UiO‑66(Ce) 892.9 m² g⁻¹ (surface area decreases with increasing pore size).
4. Chemical Valence and Surface Sites
XPS (Ce 3d): Ce³⁺ ratio increases from 36.3% (UiO‑66(Ce)) to 42.3% (2D‑mUiO‑66(Ce)), indicating more unsaturated Ce sites.
XPS (O 1s): Ce–OH proportion reaches 55.4% in 2D‑mUiO‑66(Ce), much higher than 28.5% (UiO‑66(Ce)) and 44.9% (mUiO‑66(Ce)).
XANES/EXAFS: Ce exists as mixed Ce³⁺/Ce⁴⁺; local structure is disordered with undercoordinated Ce–O bonds, consistent with XPS results.
5. Optical and Electrochemical Properties
UV–vis/ Tauc plot: Band gap 2.83 eV (2D‑mUiO‑66(Ce)), narrower than mUiO‑66(Ce) (3.00 eV) and UiO‑66(Ce) (3.07 eV).
Mott–Schottky: n‑type semiconductor; conduction band potential −0.24 V vs. Ag/AgCl (−0.04 V vs. NHE), more negative than U(VI)/U(IV) potential.
Transient photocurrent: Much higher density than reference samples, indicating fast charge transfer.
PL & EIS: Lower fluorescence intensity and smaller semicircle diameter reveal suppressed electron–hole recombination and reduced charge‑transfer resistance.
Mechanism Analysis
1. Synthesis Mechanism
PFCA forms 2D lipid bilayers that confine MOF growth into sheet morphology.
PS‑b‑PEO spherical micelles act as mesopore templates and initiate nucleation via H‑bonding between PEO and Ce precursors.
ClO₄⁻ promotes coordination between Ce clusters and BDC ligands, forming continuous 2D frameworks; template removal yields through‑mesopores.
2. Band‑Gap Modification (DFT)
Band gap follows: 3D‑UiO‑66(Ce) (3.06 eV) → 3D‑spUiO‑66(Ce) (2.91 eV) → 2D‑UiO‑66(Ce) (2.82 eV) → 2D porous models (2.79 eV).
2D morphology is the dominant factor for band‑gap narrowing; mesoporosity plays a secondary role.
3. Mass‑Transport Enhancement (MD Simulation)
Nonporous 2D‑UiO‑66(Ce) shows nearly no ion diffusion.
Large‑pore 2D‑lpUiO‑66(Ce) enables ~50% ion migration across the sheet, while small‑pore 2D‑spUiO‑66(Ce) allows only ~10–12%.
Through‑mesopores drastically shorten diffusion paths and accelerate mass transport.
4. Photocatalytic U(VI) Reduction Mechanism
Narrow band gap improves light utilization and electron–hole separation.
Negative conduction band potential provides thermodynamic driving force for U(VI) → U(IV) reduction.
Abundant Ce–OH and Ce³⁺ sites act as active centers for redox reactions.
2D mesoporous structure exposes more active sites and facilitates rapid substrate diffusion and product release.

Summary
A dual‑template interface co‑assembly method is established to synthesize ultrathin continuous 2D mesoporous UiO‑66(Ce) nanosheets with ~23 nm through‑mesopores and tunable thickness (3–150 nm).
The material exhibits substantially enhanced photocatalytic U(VI) reduction (80.3% removal) owing to improved mass transport, abundant active sites, narrowed band gap, and efficient charge separation.
DFT and MD simulations reveal that 2D morphology reduces band gap and mesopores boost ion transport, synergistically promoting catalytic performance.
Ultrathin Mesoporous Metal-Organic Framework Nanosheets
Authors: Yingji Zhao, Zhi Gao, Norman C.-R. Chen, Yusuke Asakura, Ho Ngoc Nam, Quan Manh Phung, Yunqing Kang, Mandy Hei Man Leung, Dong Jiang, Lei Fu, Lijin Huang, Toru Asahi, Yusuke Yamauchi
DOI: 10.1002/adma.202508105
Links: https://advanced.onlinelibrary.wiley.com/doi/10.1002/adma.202508105
The above review is for academic progress sharing. For any errors or copyright issues, please contact us for correction or removal.



