Home >
News > An Ultramicroporous Metal‑Organic Framework with Ultrahigh Neon Adsorption Capacity and Record Ne/He Selectivity
An Ultramicroporous Metal‑Organic Framework with Ultrahigh Neon Adsorption Capacity and Record Ne/He Selectivity
This work reports the ultramicroporous metal‑organic framework UTSA‑280, which achieves highly efficient separation of neon from helium at the industrially accessible temperature of liquid nitrogen (77 K). The material exhibits a record Ne/He selectivity of 1602 and an exceptional Ne uptake of 5.419 mmol·g⁻¹ at 77 K and 100 kPa. Breakthrough experiments and process simulations confirm its ability to produce high‑purity helium directly from a Ne/He (0.1/99.9) mixture, offering an energy‑efficient and scalable strategy for ultra‑pure helium production in electronic and cryogenic applications.
Research Background
1. Industry Problem – The removal of trace neon from helium is a critical bottleneck in producing ultrahigh‑purity (≥99.9999%) helium required for semiconductor manufacturing, cryogenics, and other advanced technologies. Conventional adsorbents (zeolites, activated carbons) suffer from either low selectivity or insufficient capacity under practically viable cryogenic conditions, often necessitating energy‑intensive processes below 50 K.
2. Existing Solutions – Commercial adsorbents show weak adsorption affinity for neon at 77 K due to neon’s low polarizability, leading to unsatisfactory capacity and selectivity. Some MOFs have demonstrated high neon adsorption under high‑pressure conditions, but systematic studies on trace‑neon removal from helium streams are lacking.
3. Innovation of This Work – The authors employ UTSA‑280, an ultramicroporous MOF with rigid one‑dimensional channels and a polarized electrostatic environment created by Ca²⁺ centers and squaric acid ligands. This design enables strong polarization and confinement of Ne atoms, achieving unprecedented Ne/He selectivity and capacity at 77 K without requiring extreme cryogenic temperatures.
Experimental Content
1. Material Synthesis – UTSA‑280 was synthesized following a reported protocol with minor modifications. The activated material was obtained by mild thermal treatment to remove guest solvent while preserving structural water molecules.
2. Thermal and Structural Characterization – Thermogravimetric analysis (TGA) showed a stable plateau between 150–250 °C, indicating good thermal stability. Powder X‑ray diffraction (PXRD) confirmed the retained crystallinity after activation. Scanning electron microscopy (SEM) revealed agglomerated hollow needle‑like crystals with smooth surfaces and high crystallinity.
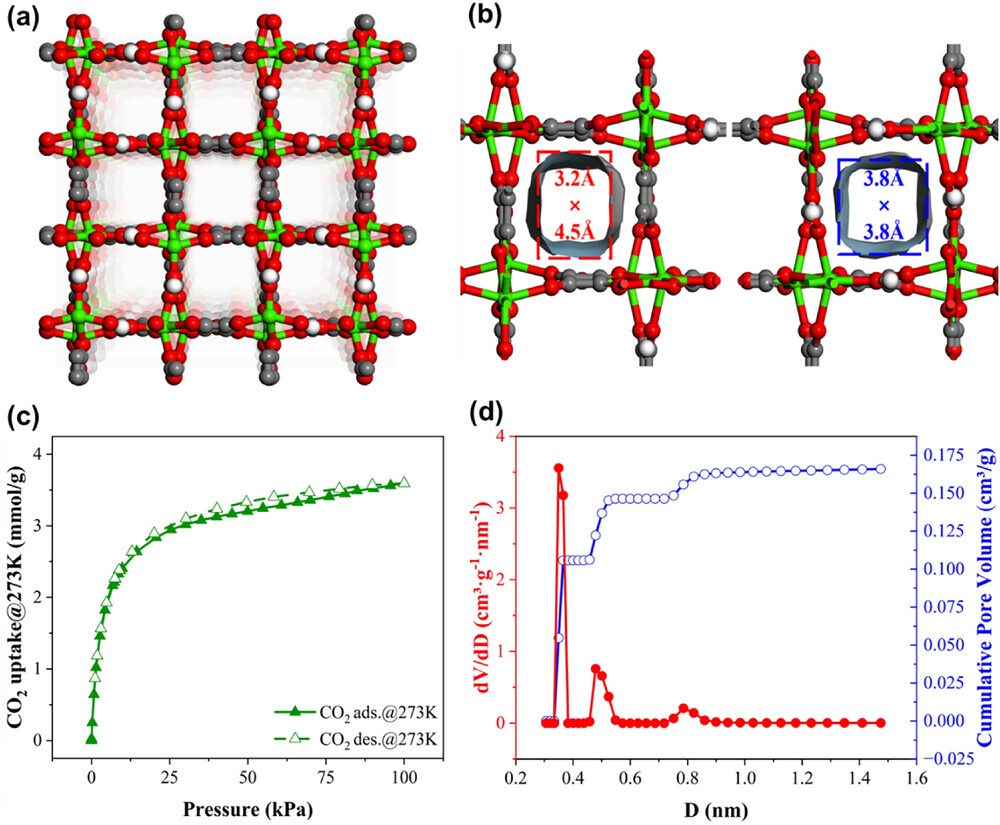
3. Gas Adsorption Measurements – CO₂ adsorption at 273 K gave a Type‑I isotherm with negligible hysteresis, confirming uniform micropores. The BET surface area calculated from CO₂ data is 464.87 m²·g⁻¹, and the pore‑size distribution (NLDFT) showed narrow peaks centered at 3.5 and 4.8 Å, matching the single‑crystal pore dimensions (3.2×4.5 and 3.8×3.8 Ų).
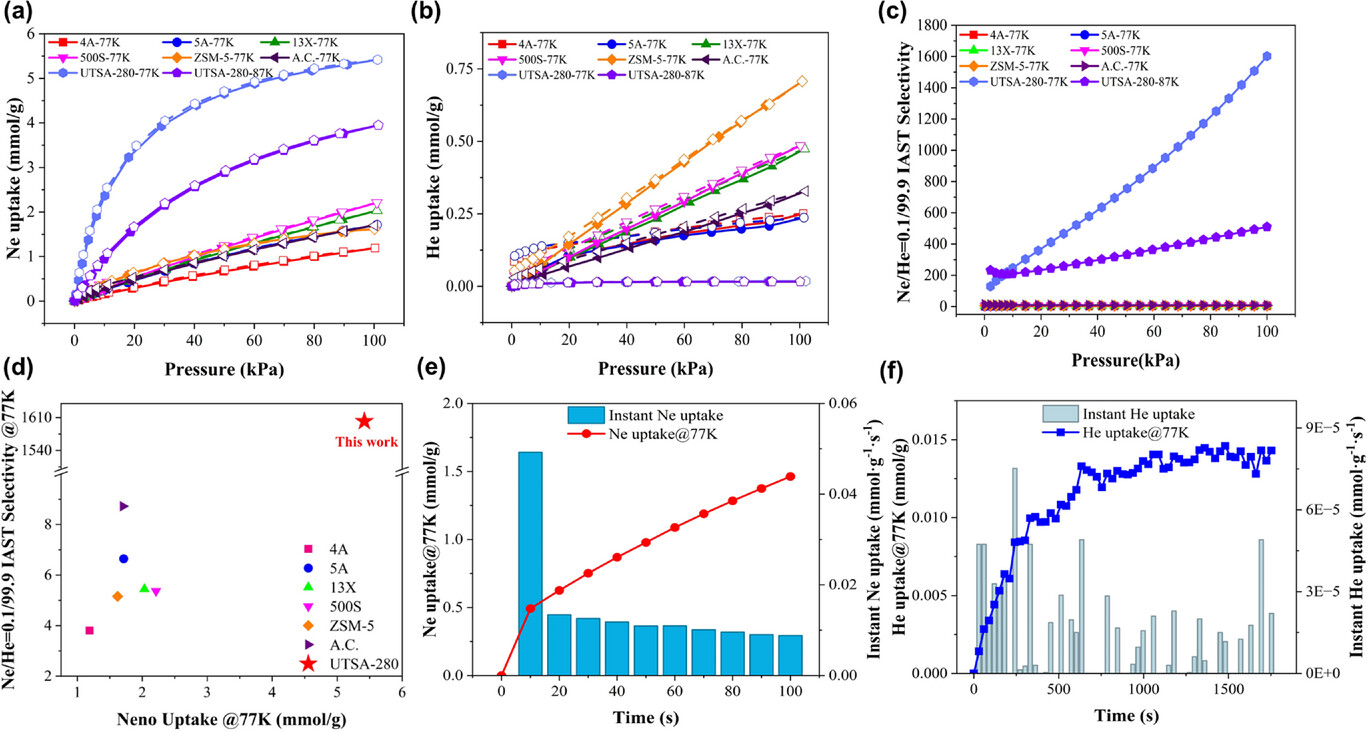
4. Single‑Gas Sorption and Selectivity – Ne and He adsorption isotherms were measured at 77 K and 87 K. UTSA‑280 delivered a Ne uptake of 5.419 mmol·g⁻¹ at 77 K and 100 kPa, more than 2.5 times that of the best commercial adsorbent (500S, 2.211 mmol·g⁻¹). He adsorption was negligible (0.018 mmol·g⁻¹). Ideal Adsorbed Solution Theory (IAST) predicted a Ne/He selectivity of 1602 for a Ne/He (0.1/99.9) mixture at 77 K, nearly two orders of magnitude higher than commercial benchmarks.
5. Adsorption Kinetics and Cyclic Stability – Ne adsorption reached an initial rate of 0.05 mmol·g⁻¹·s⁻¹, while He uptake remained extremely slow. The diffusion‑coefficient ratio of Ne to He was 50,520, indicating a powerful kinetic separation effect. Five consecutive adsorption‑desorption cycles showed fully reversible behavior with no capacity loss.
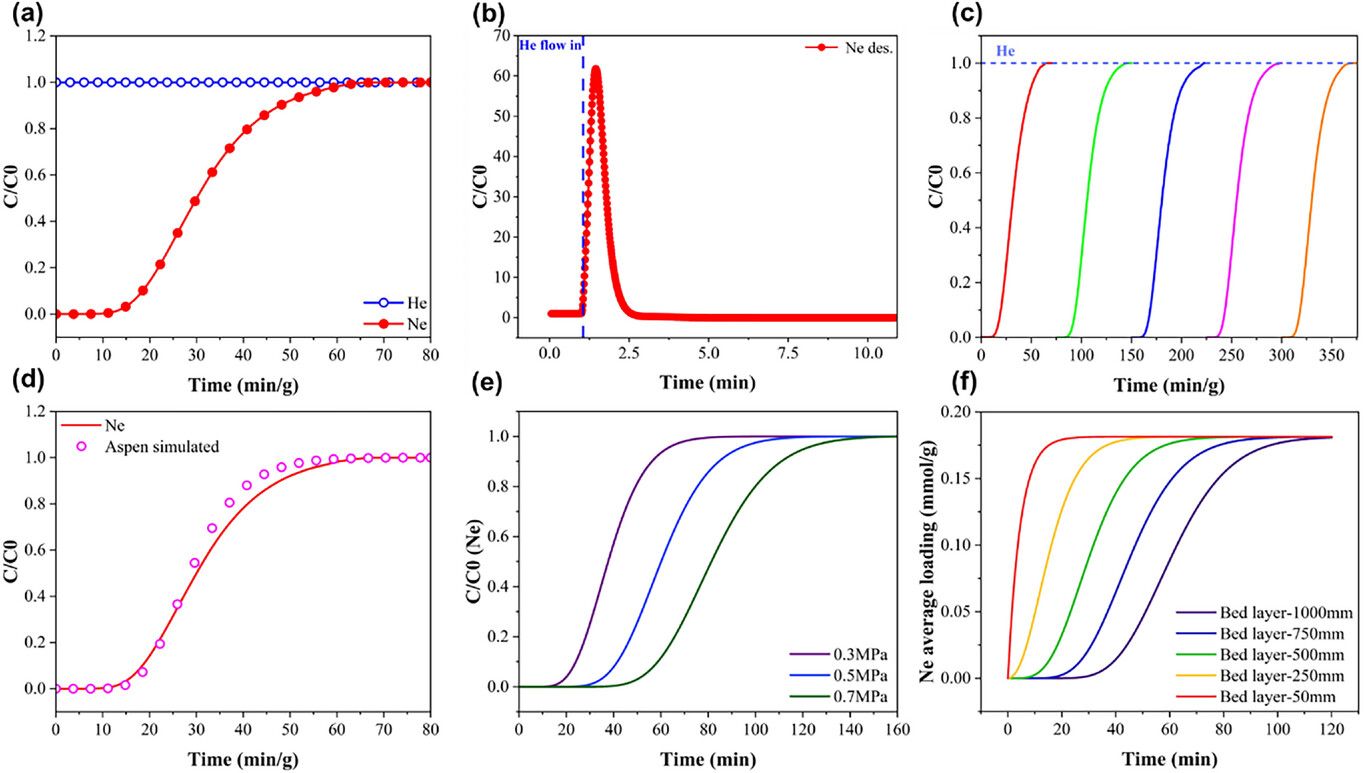
6. Breakthrough Experiments – Dynamic breakthrough tests with a Ne/He (0.1/99.9) mixture at 77 K and 100 kPa showed instantaneous He breakthrough and strong Ne retention. The breakthrough point (C/C₀ = 0.05) and saturation point (C/C₀ = 0.95) were delayed to 16.63 and 54.84 min·g⁻¹, respectively. Regeneration with pure He at 298 K rapidly desorbed Ne, and five consecutive cycles confirmed excellent cyclic stability.
7. Process Simulation – A fixed‑bed adsorption model built in Aspen Adsorption was validated with experimental data. Simulations at an industrial scale (200 mm × 1000 mm column) indicated that an operating pressure of 0.5 MPa provides a balanced breakthrough time (37.28 min) and economic efficiency.
Characterization and Analysis
·Surface Area and Porosity – BET surface area: 464.87 m²·g⁻¹; micropore volume: 0.16 cm³·g⁻¹; pore‑size distribution: narrow peaks at 3.5 and 4.8 Å.
·Single‑Gas Uptake – Ne capacity: 5.419 mmol·g⁻¹ at 77 K, 100 kPa; He capacity: 0.018 mmol·g⁻¹ under the same conditions.
·Selectivity – IAST‑predicted Ne/He selectivity: 1602 at 77 K for Ne/He (0.1/99.9).
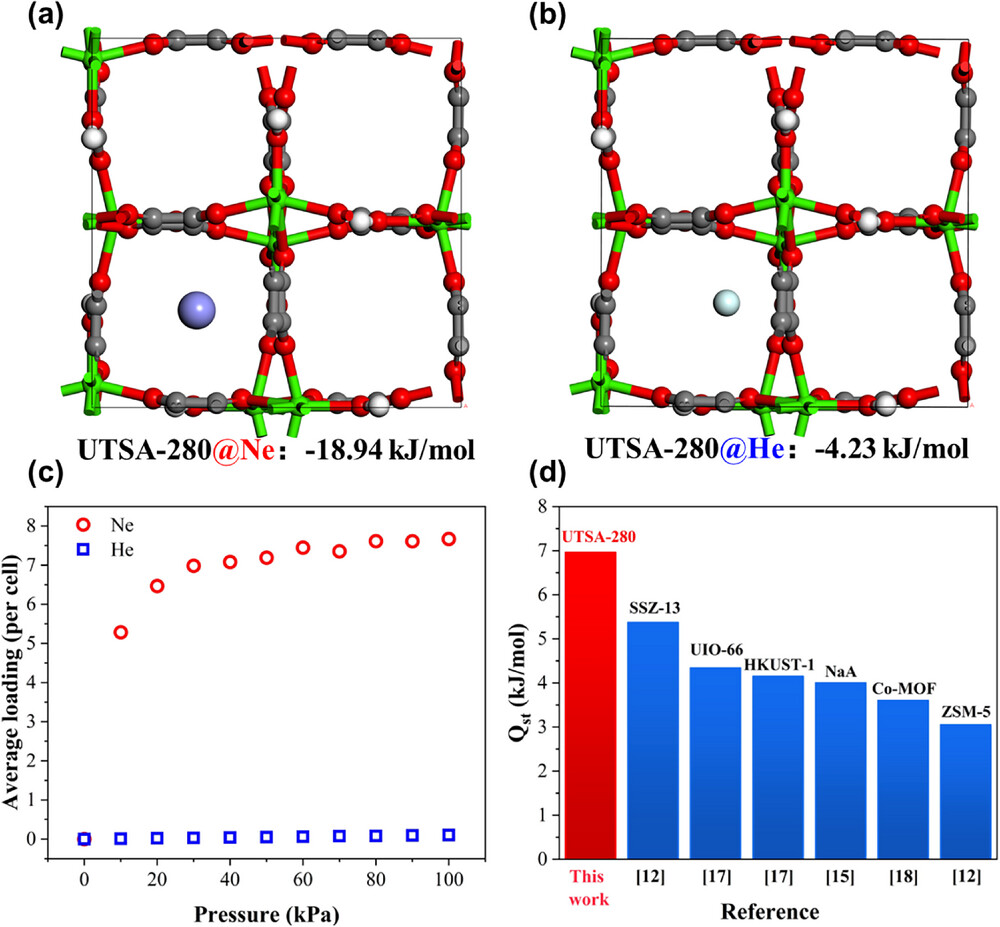
·Adsorption Heat – Isosteric heat of adsorption (Qst) for Ne: 6.96 kJ·mol⁻¹, the highest value reported to date for neon adsorbents.
·Breakthrough Performance – He broke through instantly; Ne breakthrough delayed to 16.63 min·g⁻¹ (C/C₀ = 0.05) and saturation at 54.84 min·g⁻¹ (C/C₀ = 0.95).
·Cyclic Stability – Five consecutive breakthrough‑regeneration cycles showed nearly identical curves, confirming robust long‑term performance.
Mechanism Analysis
The authors employed density‑functional theory (DFT), Mulliken charge analysis, and grand‑canonical Monte Carlo (GCMC) simulations to elucidate the separation mechanism.
1. DFT Calculations – The binding energy of Ne in UTSA‑280 is significantly lower than that of He, attributed to the polarization of Ne atoms by the electrostatic field generated by Ca²⁺ centers and squaric acid ligands.
2. Mulliken Charge Analysis – Ne acquired an atomic charge of 0.05312, substantially higher than that of He (0.00615), confirming stronger electrostatic interactions.
3. GCMC Simulations – Although He atoms can access more positions in the channels, their probability density at the channel center is about two orders of magnitude lower than that of Ne. At 0.1 bar and 77.3 K, the average unit‑cell loading of Ne (5.284 molecules per unit cell) exceeds that of He (~0.01) by two orders of magnitude.
4. Adsorption Heat – The high Qst (6.96 kJ·mol⁻¹) indicates strong polarization and confinement effects on Ne atoms, explaining the thermodynamic origin of the superior separation performance.
Summary
Work Summary – The authors demonstrate that UTSA‑280, an ultramicroporous Ca‑based MOF with squaric acid ligands, achieves a record Ne/He selectivity of 1602 and a high Ne uptake of 5.419 mmol·g⁻¹ at 77 K. Breakthrough experiments and process simulations validate its ability to produce high‑purity helium from a trace‑neon mixture, offering a practical, energy‑efficient alternative to conventional cryogenic processes.
A Ultramicroporous Metal-Organic Framework With Ultrahigh Neon Adsorption Capacity and Record Ne/He Selectivity
Authors: Hedong Yang, Yinhui Li, Qingkuan Meng, Yongzheng Wang, Zhihao Bian, Zhizheng Wang, Heping Ma
DOI: 10.1002/adfm.75261
Links: https://advanced.onlinelibrary.wiley.com/doi/10.1002/adfm.75261
The above review is for academic progress sharing. For any errors or copyright issues, please contact us for correction or removal.



