Home >
News > Nanoconfinement-enabled selective trapping of trace propane in a shaped Al-MOF for ultra-pure fluoromethane
Nanoconfinement-enabled selective trapping of trace propane in a shaped Al-MOF for ultra-pure fluoromethane
Ultra-high-purity fluoromethane (CH₃F) is a vital electronic special gas for semiconductor manufacturing, yet deep purification is hindered by trace propane (C₃H₈) impurities. This work develops a molded aluminum-based metal–organic framework (Al-Fum) with precisely tailored 5.6 × 6.0 Å one-dimensional channels. By leveraging polarizability-driven nanoconfinement, the weakly polar Al-Fum selectively amplifies van der Waals interactions with highly polarizable C₃H₈ while weakening binding to weakly polar CH₃F. The adsorbent reduces propane concentration from 100 ppm to the parts-per-trillion level, achieving 6N/7N-purity CH₃F production. Scalable wet extrusion molding preserves the material’s intrinsic performance, with pellets showing high mechanical strength and excellent regenerability. This study clarifies the nanoconfinement separation mechanism and provides a practical solution for industrial electronic gas purification.
Research Background
1. Industrial and Technical Challenges
The semiconductor industry demands ultra-high-purity CH₃F for thin-film processes, but trace C₃H₈ (≈100 ppm) degrades device performance. Cryogenic distillation for deep removal is energy-intensive and economically unviable.
CH₃F and C₃H₈ have similar kinetic diameters (3.8 Å vs. 4.3–5.1 Å), making simple size-sieving ineffective and requiring thermodynamic selectivity-based strategies.
2. Existing Solutions and Limitations
Traditional adsorbents (amorphous carbons, zeolites) lack molecular-level pore tunability, showing low C₃H₈ uptake and modest C₃H₈/CH₃F selectivity.
Reported porous carbons (e.g., CMS-600) fail to meet ultra-purification requirements, with low propane uptake and limited 6N-purity CH₃F productivity.
Most MOFs are in powder form; molding typically causes severe porosity/active site loss, hindering industrial translation.
3. Work Innovations
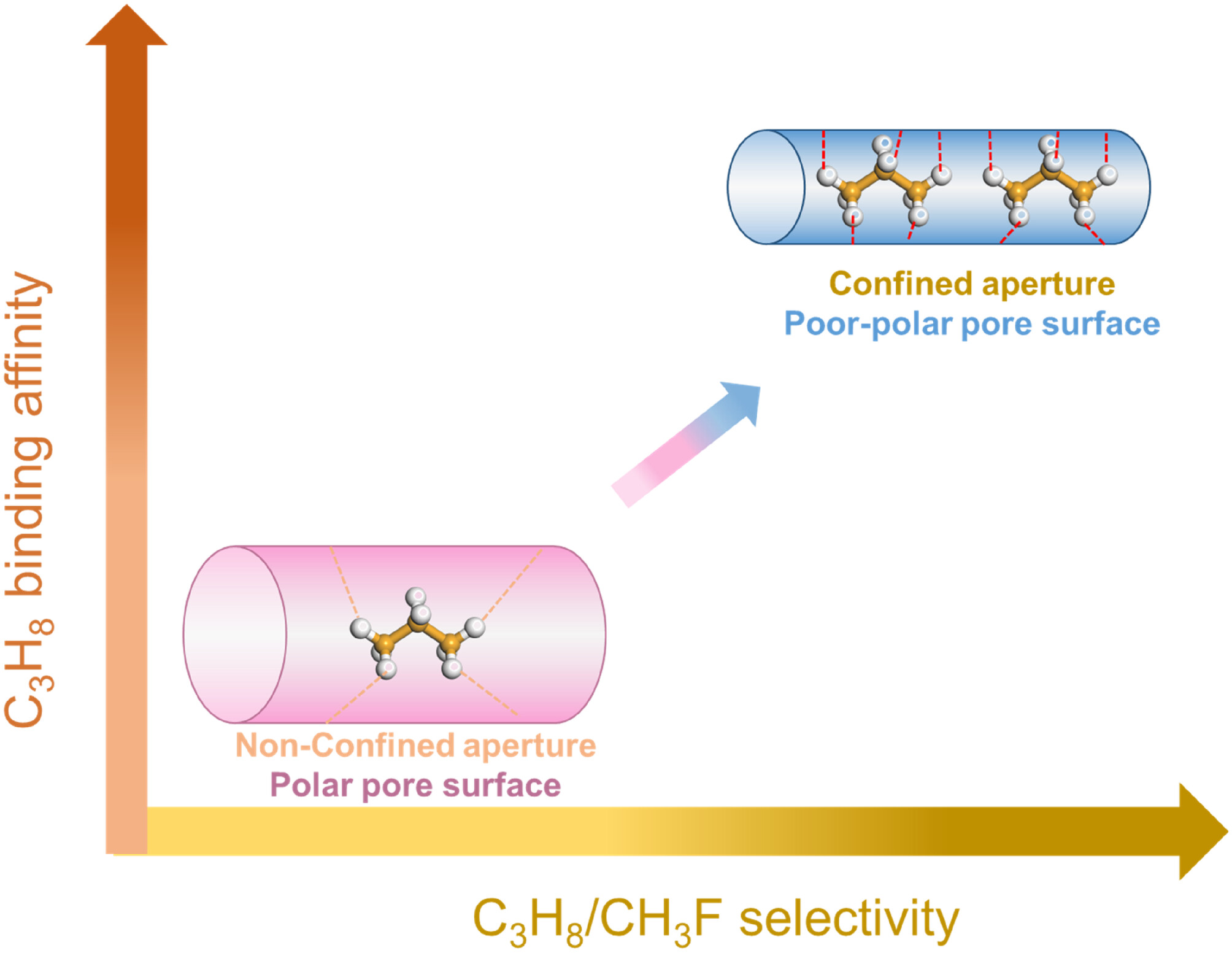
Propose a polarizability-driven nanoconfinement strategy to selectively enhance C₃H₈ adsorption via tailored pore environments.
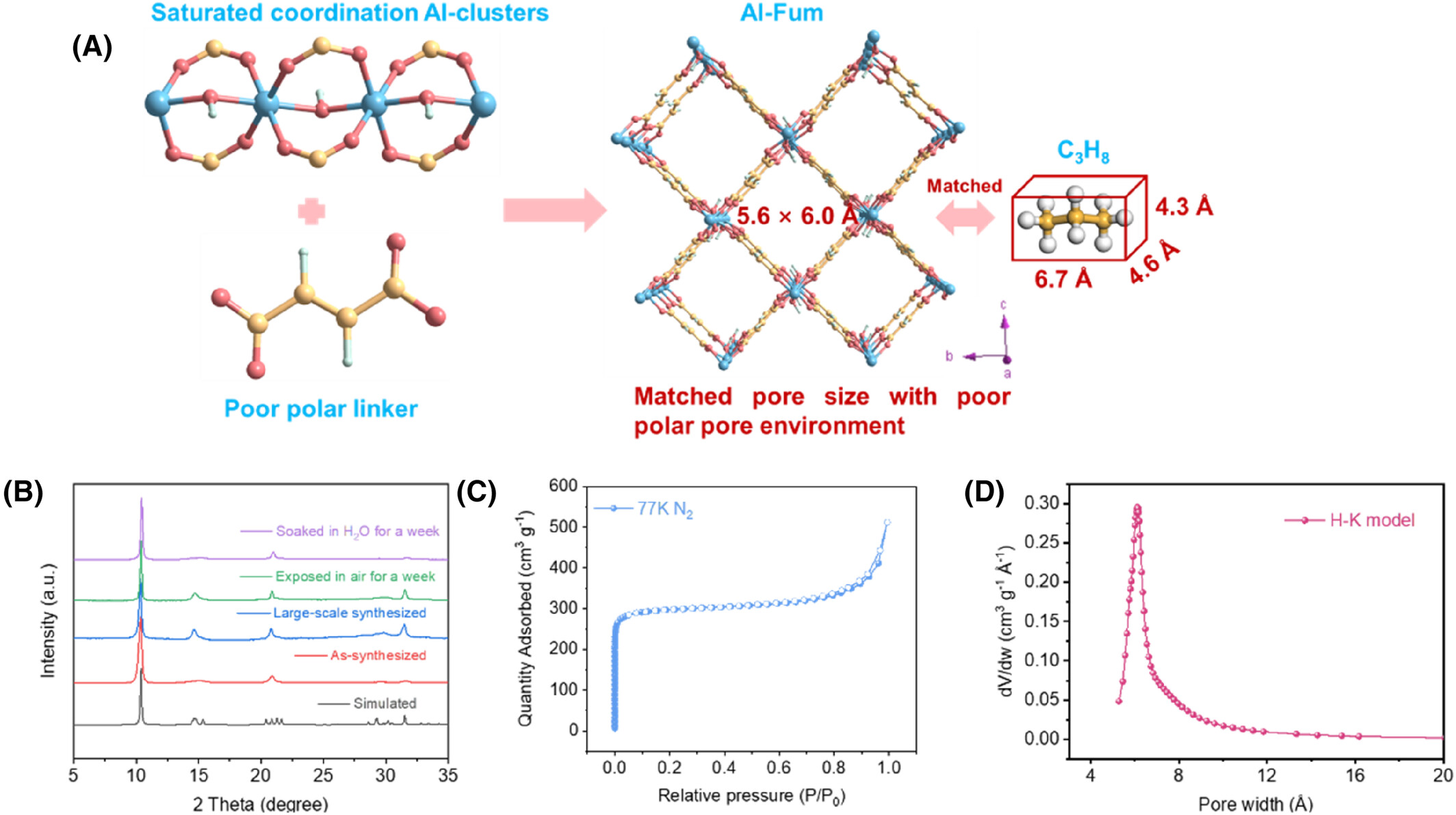
Synthesize stable Al-Fum with 5.6 × 6.0 Å channels, perfectly matching C₃H₈ molecular size.
Develop a scalable wet extrusion molding method balancing mechanical strength and adsorption performance.
Achieve sub-ppb-level C₃H₈ removal, with 6N/7N-purity CH₃F productivity exceeding all reported benchmarks.
Experimental Content
1. Material Synthesis & Molding
Al-Fum powder was synthesized via a green, solvent-free aqueous stirring method at 333 K. Four binders (CM, HEC, HPMC, PVA) at 2.5–10 wt% were used to prepare cylindrical pellets (0.5–1 cm) via wet extrusion.
2. Single-Component Adsorption
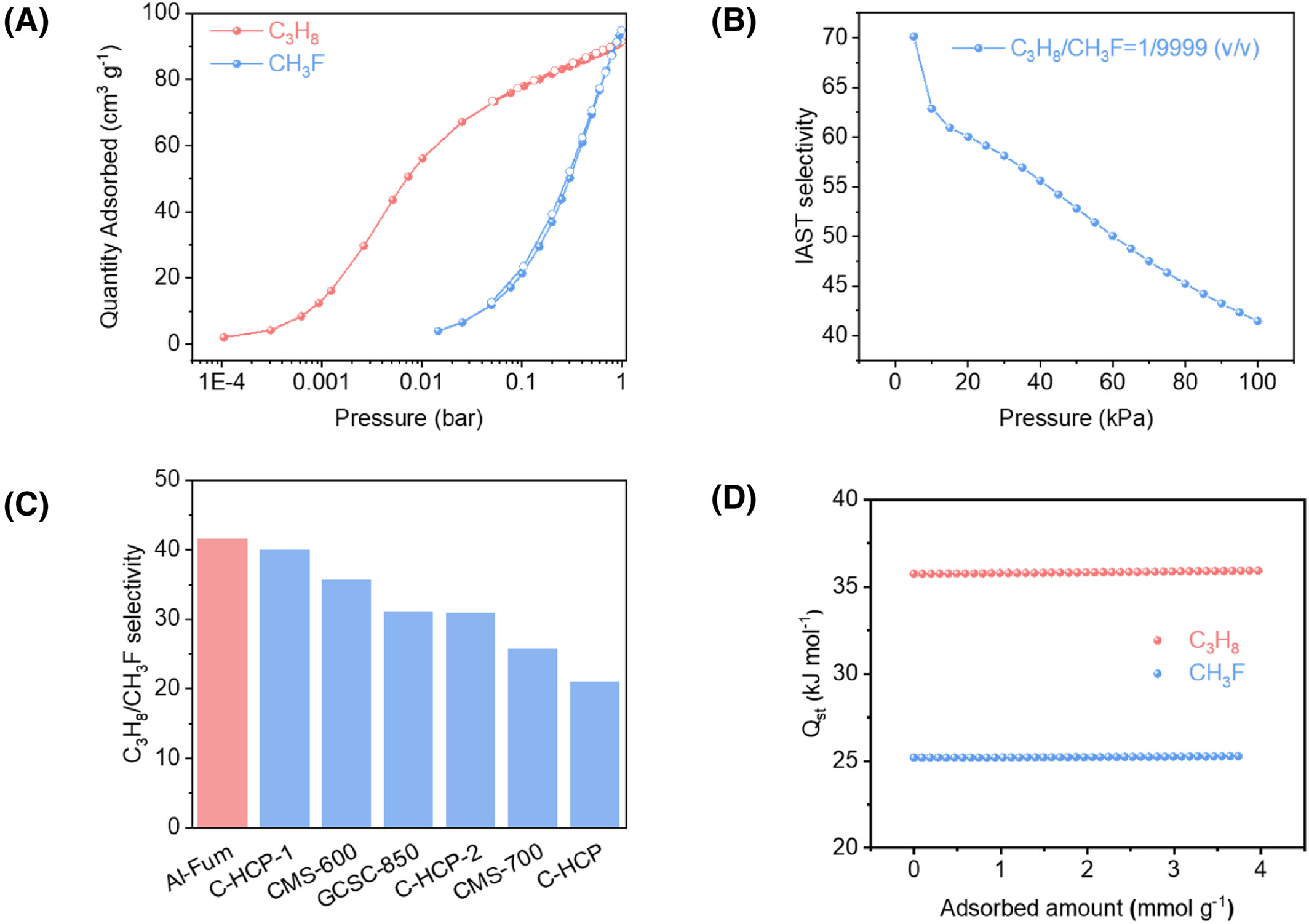
C₃H₈ adsorption isotherms at 298 K showed sharp low-pressure uptake: 2.06 cm³ g⁻¹ at 0.0001 bar (100 ppm), far exceeding commercial CTC-100/CMS-600 (<1 cm³ g⁻¹). Saturated C₃H₈ capacity at 1 bar was 91.53 cm³ g⁻¹, vs. 94.82 cm³ g⁻¹ for CH₃F. IAST C₃H₈/CH₃F (1/9999) selectivity reached 41.6, surpassing C-HCP-1 (40), CMS-600 (35), and GCSC-850 (31).
3. Dynamic Breakthrough Tests
For 1/9999 C₃H₈/CH₃F (10 mL min⁻¹, 298 K, 1 bar), C₃H₈ retention time on Al-Fum was 306 min·g⁻¹ (vs. 135 min·g⁻¹ for CTC-100). 6N-purity CH₃F productivity exceeded 300 min·g⁻¹; five cycles showed no performance decay. 6N CH₃F productivity (2994 L·kg⁻¹) outperformed C-HCP-2 (2843 L·kg⁻¹), GCSC-900 (~2011 L·kg⁻¹), CTC-100 (1180 L·kg⁻¹), and CMS-600 (457 L·kg⁻¹).
4. Molding Performance
All pellets had crushing strength >20 N/particle (vs. 11.07 N/particle for carbon). Al-Fum@2.5%CM/PVA retained >98% C₃H₈ capacity, with strengths of 22.6 N/particle and 34.57 N/particle. Al-Fum@2.5%CM produced 7N-purity CH₃F for 219.3 min·g⁻¹ (2193 L·kg⁻¹).
Characterization and Analysis
1. Structural Characterization
PXRD confirmed Al-Fum’s crystallinity, retained after synthesis, air exposure (1 week), and water soaking (1 week). N₂ adsorption–desorption (77 K) gave a Type-I isotherm with BET surface area = 1139 m² g⁻¹. DFT/H-K pore size distribution showed a dominant peak at ~5.8 Å, matching crystallographic 5.6 × 6.0 Å channels.
Crystal structure: Infinite AlO₆ chains connected by low-polarity fumarate ligands form uniform 1D channels.
2. Adsorption Thermodynamics
Isosteric heat of adsorption (Qst): C₃H₈ = 35.2 kJ·mol⁻¹, CH₃F = 25.1 kJ·mol⁻¹, confirming stronger C₃H₈–host interactions.
3. DFT Results
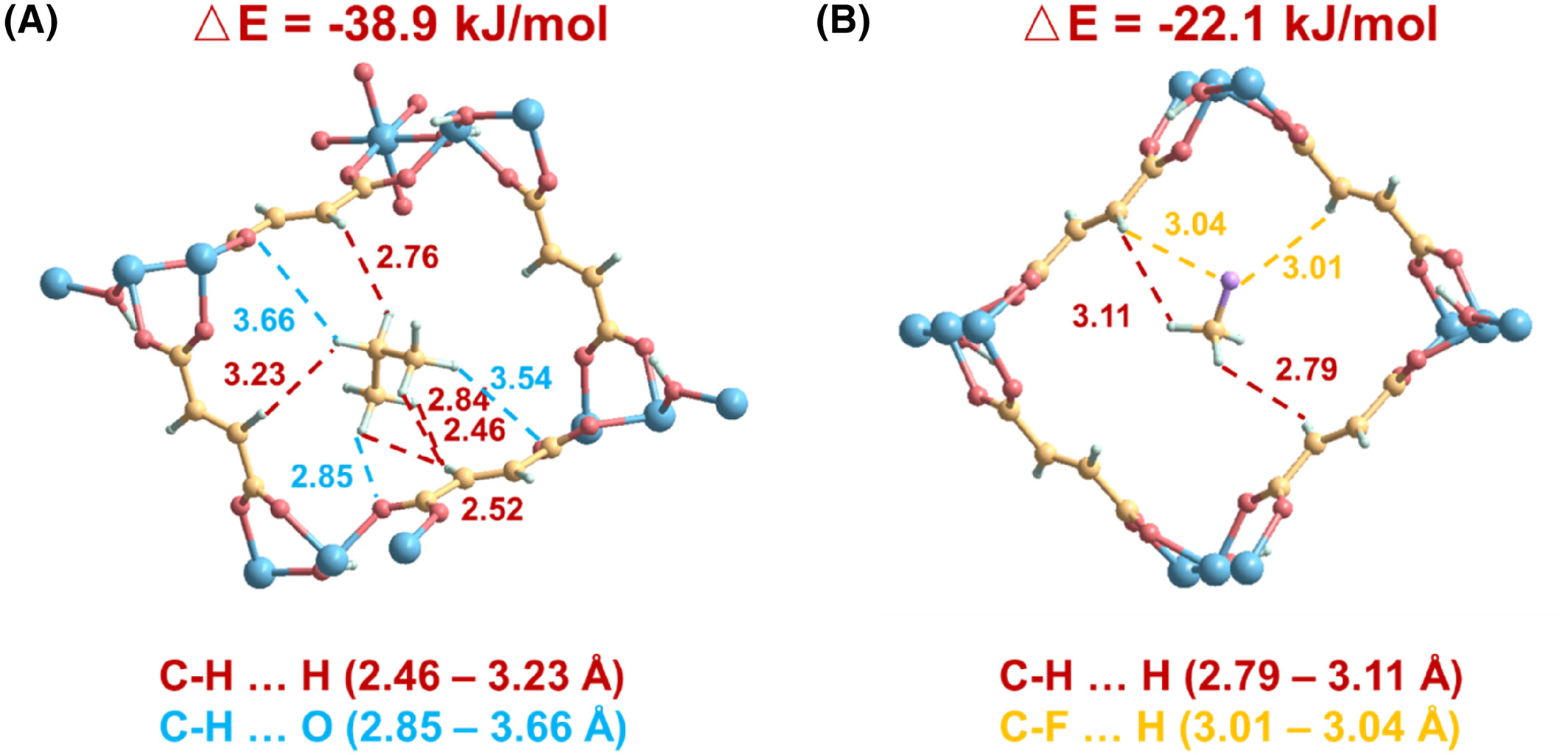
Binding energies: C₃H₈ = −38.9 kJ·mol⁻¹, CH₃F = −22.1 kJ·mol⁻¹ (consistent with Qst). C₃H₈ forms strong C–H…π (2.47–3.23 Å) and C–H…O (2.85–3.66 Å) contacts; CH₃F only weak C–H…H/C–F…H interactions.
4. Molding Characterization
Molded pellets had smooth surfaces and retained BET/pore structure. Breakthrough curves matched powder, confirming preserved separation efficiency.
Mechanism Analysis
1. Nanoconfinement Effect
Al-Fum’s 5.6 × 6.0 Å channels slightly exceed C₃H₈’s kinetic diameter, imposing spatial confinement that increases van der Waals contact between C₃H₈ and the channel wall.
2. Polarizability-Driven Selectivity
C₃H₈ (polarizability ≈6.3 × 10⁻²⁴ cm³) has far higher polarizability than CH₃F (≈2.5 × 10⁻²⁴ cm³). Under the channel’s electric field gradient, C₃H₈ forms a stronger induced dipole. The weakly polar channel surface minimizes electrostatic attraction to CH₃F’s permanent dipole (1.85 D), maximizing selectivity.
3. Binding Interaction Differences
C₃H₈ is stabilized by multiple strong van der Waals interactions; CH₃F only forms weak non-directional forces. Moderate Qst (35.2 kJ·mol⁻¹) enables energy-efficient regeneration (vacuum at 393 K for 2 h).
4. Structure–Performance Link
Coordinatively saturated Al sites and low-polarity ligands avoid strong CH₃F binding, while 1D channels facilitate fast mass transfer for high productivity.
Summary
1. Developed Al-Fum as a high-performance adsorbent for deep removal of trace C₃H₈ from CH₃F, based on a novel polarizability–nanoconfinement synergistic mechanism.
2. Achieved ultra-deep purification: reduced C₃H₈ from 100 ppm to sub-ppb level, realizing one-step production of 6N and 7N electronic-grade fluoromethane.
3. Established a scalable extrusion molding process; optimized Al-Fum@2.5%CM pellets retain >98% adsorption capacity with high mechanical strength, enabling industrial application.
4. Verified excellent stability, water resistance, and regenerability through dynamic cycling and structural tests, with performance exceeding all reported benchmark materials.
Nanoconfinement-enabled selective trapping of trace propane in a shaped Al-MOF for ultra-pure fluoromethane
Authors:Jinze Yao, Siyao Zhao, Dongyu Chen, Wenxuan Lin, Xiaofei Chen, Zehan Li, Liangxin Ding, Zhong Li, Qibin Xia
DOI: 10.1002/aic.70381
Links: https://aiche.onlinelibrary.wiley.com/doi/10.1002/aic.70381
The above review is for academic progress sharing. For any errors or copyright issues, please contact us for correction or removal.



