Home >
News > High-throughput Screening-guided Discovery of Stable Metal-Organic Frameworks for Efficient Ambient Nitrogen Dioxide Removal
High-throughput Screening-guided Discovery of Stable Metal-Organic Frameworks for Efficient Ambient Nitrogen Dioxide Removal
Nitrogen dioxide (NO₂) is a highly toxic air pollutant that poses a severe threat to the sustainable control of air pollution, and the efficient capture of NO₂ under ambient conditions remains a major technical challenge. In this study, the researchers combined a NO₂-specific high-throughput computational screening (HTCS) of more than 15,000 metal-organic frameworks (MOFs) from the CoRE database with targeted experimental validation to screen out stable aluminum-based MOFs (Al-MOFs) for selective NO₂ capture. Guided by the physicochemical properties of NO₂ and the principles of synthetic feasibility, four optimized Al-MOFs (KMF-1, CAU-23, MIL-160 and MOF-303) with different heteroatom functional sites (-NH, -S, -O and -N-NH respectively) were selected to investigate the structure-adsorption correlation. Among them, MOF-303 exhibited an outstanding dynamic NO₂ adsorption capacity of 5.31 mmol·g⁻¹, which exceeded most of the reported porous adsorbents. Spectroscopic analysis and density functional theory (DFT) calculations revealed that the synergistic dipole interaction and hydrogen bonding at the unique -N-NH bifunctional sites dominated the NO₂ adsorption behavior of MOF-303. Such site-specific interactions endowed MOF-303 with excellent long-term stability and regenerability, which also verified the rationality of the physicochemical descriptor-guided screening strategy and confirmed its reliability under practical application conditions. Overall, this study connects theoretical prediction with experimental verification, and establishes a transferable research paradigm for targeted environmental remediation and the screening of next-generation functional materials.
Research Background
1. Existing problems in the field:
NO₂ is a toxic air pollutant that endangers the environment and human health, with the US CDC setting 20 ppm as its immediately dangerous to life and health (IDLH) threshold. Conventional NO₂ removal technologies such as selective catalytic reduction (SCR) and non-selective catalytic reduction (SNCR) operate at high temperatures (above 473 K), making them unsuitable for low-temperature indoor applications and energy-efficient deployment. Meanwhile, the highly oxidative and acidic nature of NO₂ easily damages the structural integrity of porous adsorbents, and the lack of systematic large-scale experimental screening methods and the poor scalability of high-performance MOFs restrict the development of practical NO₂ adsorbents.
2. Solutions proposed by other researchers:
Porous materials based on physical and chemical adsorption have attracted extensive attention for passive and scalable NO₂ mitigation, and MOFs show unique advantages due to their tunable pore environment, high specific surface area and modular chemical structure. Microporous MOFs are suitable for adsorbing small and reactive NO₂ molecules, and researchers have explored moderate interaction mechanisms such as dipole-dipole alignment and hydrogen bonding to enhance NO₂ capture while maintaining structural stability, and functional groups containing heteroatoms (N, O, S) can mediate such interactions. However, traditional MOF discovery methods rely on trial-and-error synthesis, which is time-consuming and resource-intensive, and previous high-throughput computational screening only considered geometric factors, ignoring the physicochemical properties of NO₂ and the synthetic feasibility of materials.
3. New ideas and innovative improvements of the authors:
The research team developed a NO₂-specific HTCS strategy guided by molecular-level physicochemical descriptors, which integrated pore accessibility, framework polarity and acid-resistant robustness (parameters directly related to NO₂'s size, polarity and corrosivity) into the screening criteria, breaking the limitation of single geometric factor screening in previous studies. On this basis, the authors further added a synthetic feasibility analysis to screen MOFs with economic efficiency, scalability and environmental compatibility for experimental validation. In addition, the team selected four Al-MOFs with different heteroatom functional sites to systematically explore the structure-performance correlation of MOFs for NO₂ adsorption, and combined multiple characterization techniques and theoretical calculations to elucidate the molecular-level adsorption mechanism, establishing a closed-loop design rationale from theoretical prediction to experimental verification and mechanism analysis.
Experimental Content
1. High-throughput computational screening of MOFs:
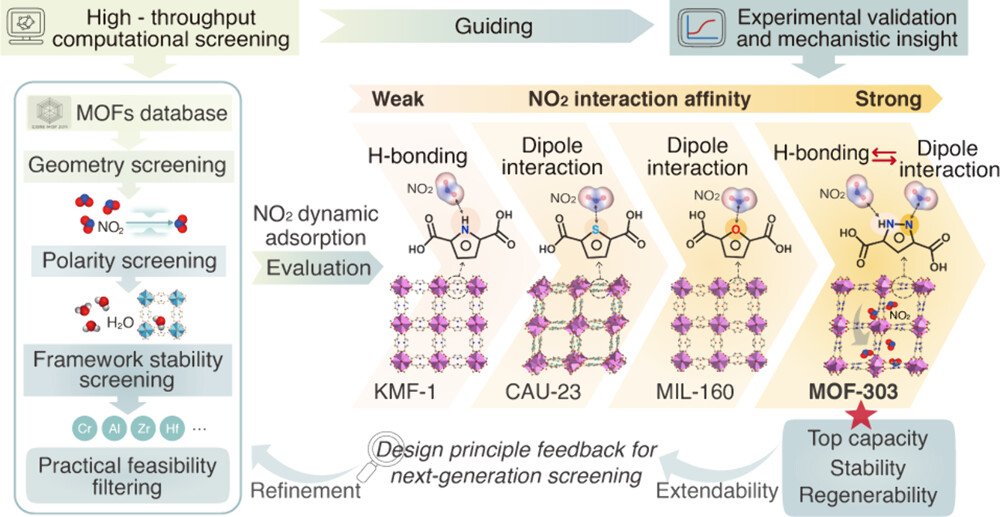
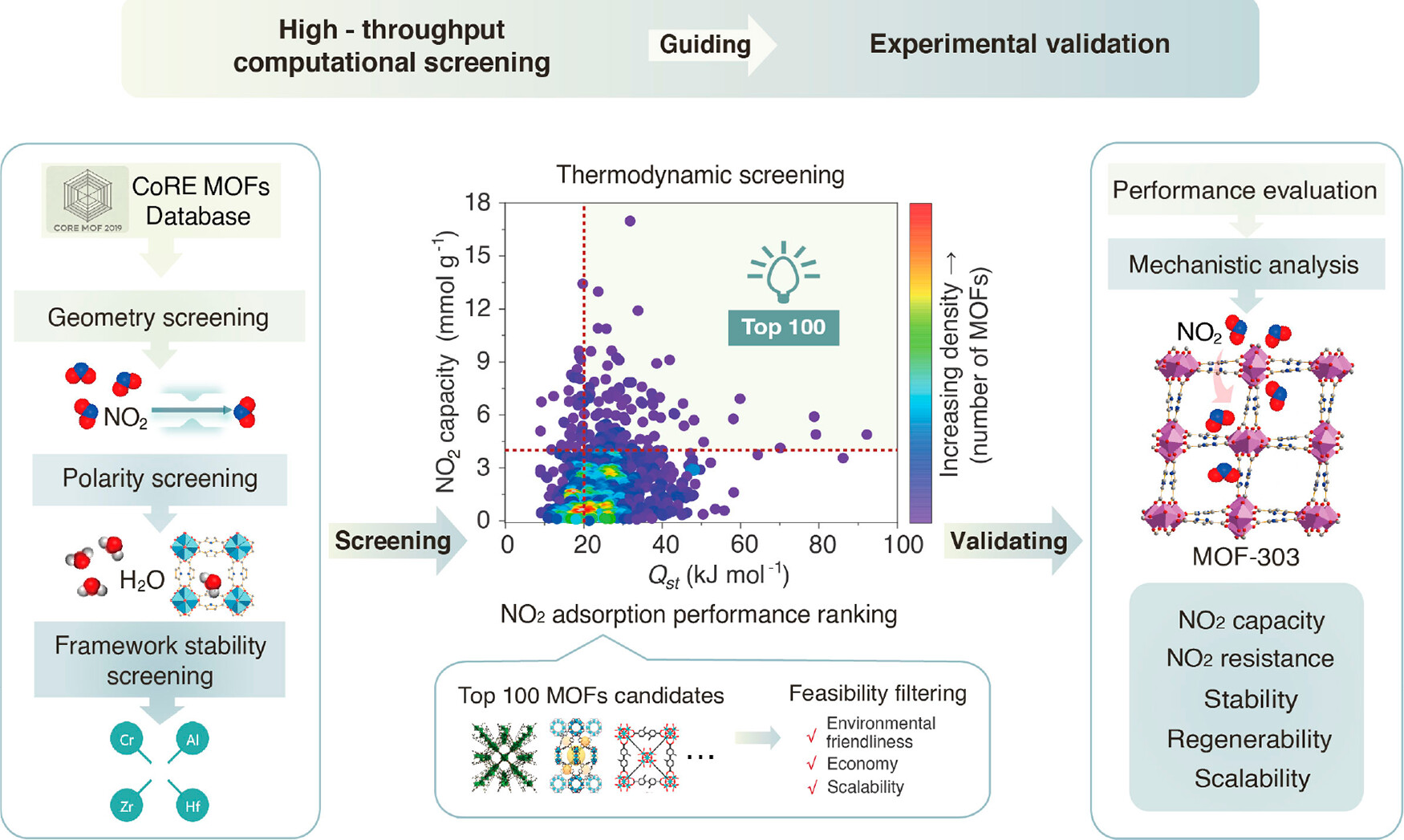
The authors applied NO₂-specific screening filters to 15,337 MOFs from the CoRE MOF 2019-2023 dataset based on grand canonical Monte Carlo (GCMC) simulations, and designed four sequential screening criteria according to the physicochemical properties of NO₂: (1) pore-limiting diameter > 3.3 Å to ensure NO₂ diffusion accessibility; (2) hydrophilicity index as a polarity descriptor to provide moderate dipole interaction; (3) prioritizing high-valence metal nodes (Al, Zr, Cr, Hf) based on the hard-soft acid-base (HSAB) theory to ensure framework acid resistance; (4) thermodynamic evaluation correlating simulated NO₂ capacity with isosteric heat of adsorption (Qst). This screening workflow identified 100 MOFs with optimal trade-offs between adsorption strength and framework stability, and further narrowed down the candidates by evaluating organic linker cost, solvent toxicity and scalability, finally selecting KMF-1, CAU-23, MIL-160 and MOF-303 for experimental validation.
2. Synthesis of Al-MOFs:
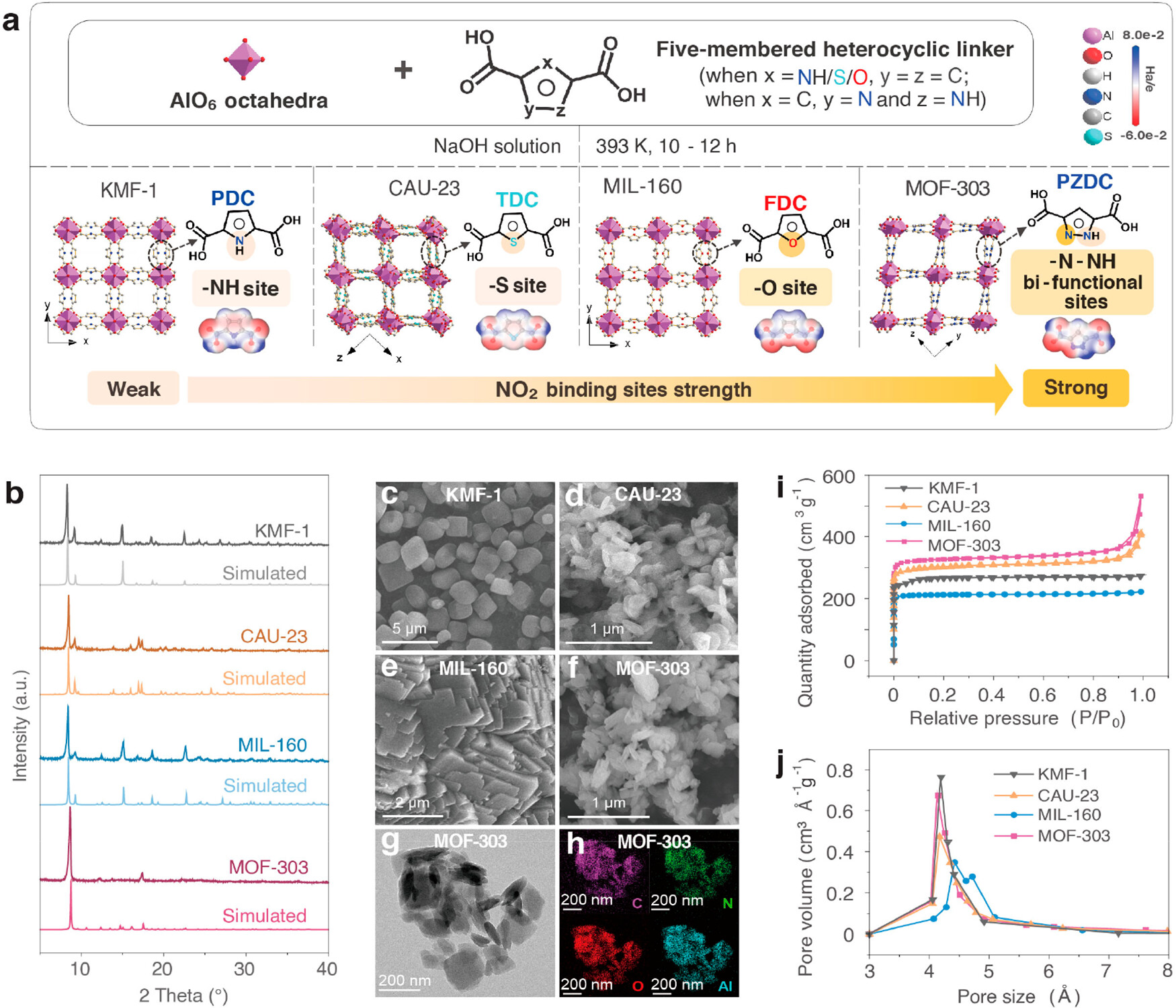
The four Al-MOFs were synthesized with Al as the metal node and different five-membered heterocyclic linkers: KMF-1 (Al-PDC, PDC = ¹H-pyrrole-2,5-dicarboxylate) with -NH sites, CAU-23 (Al-TDC, TDC = 2,5-thiophene dicarboxylate) with -S sites, MIL-160 (Al-FDC, FDC = 2,5-furan dicarboxylate) with -O sites, and MOF-303 (Al-PZDC, PZDC = 3,5-pyrazole dicarboxylate) with -N-NH bifunctional sites. The synthesis used low-cost commercial precursors and non-toxic solvents, and MOF-303 achieved a yield of more than 90% at the 100 g scale, showing excellent scalability.
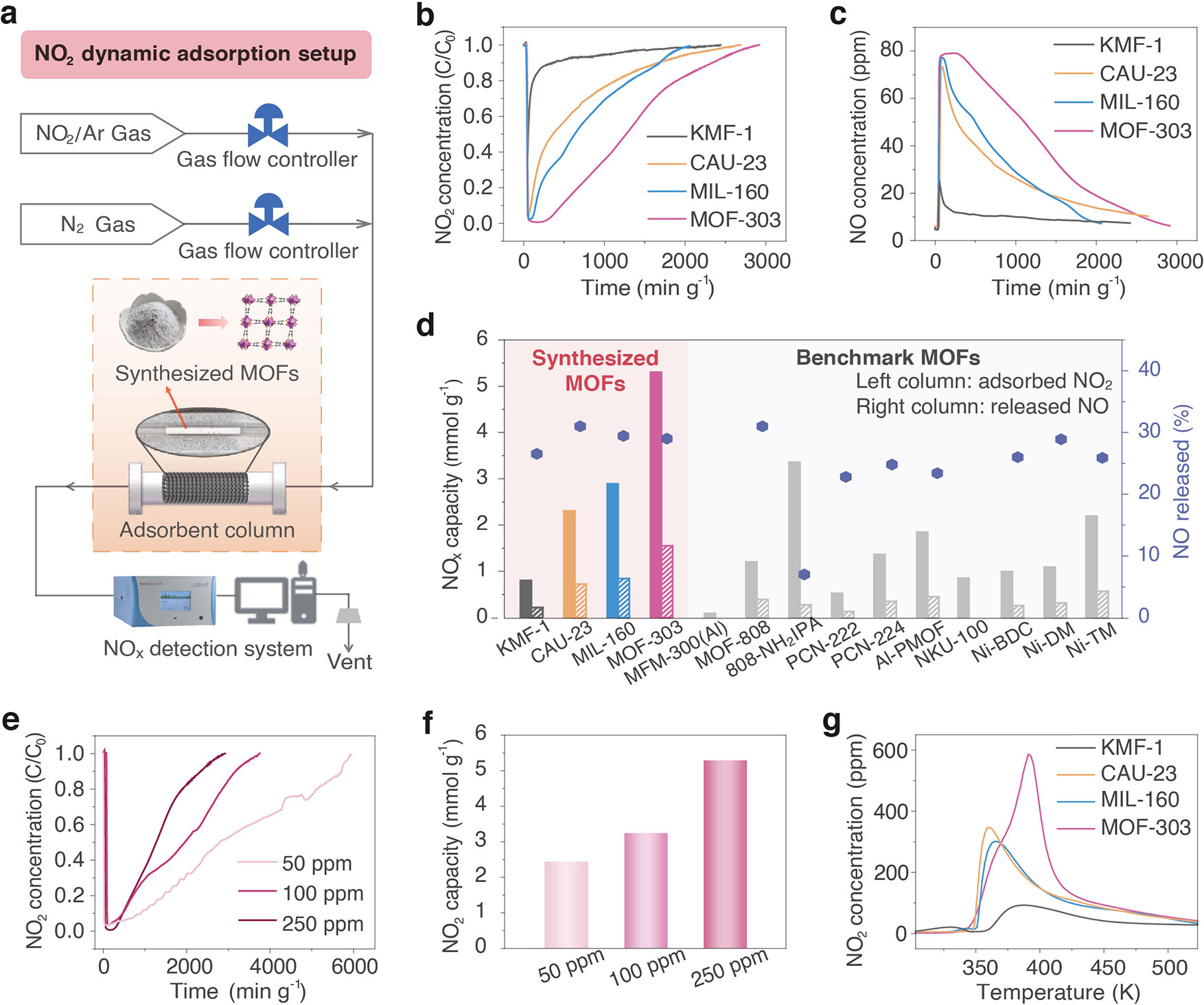
3. Dynamic NO₂ adsorption experiments:
A continuous-flow fixed-bed experimental setup was constructed to simulate realistic indoor NO₂ removal conditions, and the dynamic NO₂ adsorption performance of the four Al-MOFs was tested at atmospheric pressure with a NO₂/N₂ mixture (250 ppm, higher than typical indoor levels for accurate quantification). The results showed that MOF-303 had the highest adsorption capacity (5.31 mmol·g⁻¹), followed by MIL-160 (2.90 mmol·g⁻¹), CAU-23 (2.31 mmol·g⁻¹) and KMF-1 (0.81 mmol·g⁻¹). MOF-303's capacity significantly exceeded that of reported benchmark adsorbents (e.g., activated carbon GAC-O-T (2.96 mmol·g⁻¹), zeolite Cu²⁺SSZ-13-190-R (1.79 mmol·g⁻¹), MOF NKU-100 (0.86 mmol·g⁻¹)). In addition, the adsorption capacity of MOF-303 at 50 ppm and 100 ppm NO₂ was tested to be 2.49 mmol·g⁻¹ and 3.27 mmol·g⁻¹ respectively, simulating low-concentration indoor conditions.
4. Regeneration and practical performance tests of MOF-303:
The regenerability of MOF-303 was evaluated by cyclic adsorption-desorption experiments: the first four cycles used deionized water washing combined with thermal activation (393 K, vacuum), and the fifth cycle used mild alkaline solution washing (NaOH, pH ≈12). Breakthrough experiments were conducted using a breakthrough analyzer-mass spectrometer (BTAMS) to test the NO₂ adsorption performance of MOF-303 under dry and humid (50% RH) conditions, and the applicability of MOF-303 in different practical scenarios (kitchen range hood, parking garage, metro station) was modeled based on typical indoor NO₂ concentrations (2-20 ppb).
5. Temperature-programmed desorption (TPD) experiments:
TPD experiments were carried out on the four Al-MOFs after dynamic adsorption to analyze their NO₂ adsorption-desorption behavior. The results showed that MOF-303 had a prominent desorption peak at 396 K (the highest temperature), MIL-160 at 363 K, CAU-23 at 348 K, and KMF-1 only had a broad low-intensity peak at about 383 K, reflecting the difference in physisorption strength of different heteroatom functional sites to NO₂.
Characterization and Analysis
1. Structural and morphological characterization:
Powder X-ray diffraction (PXRD) patterns of the four synthesized Al-MOFs were consistent with the simulated patterns from the CCDC database, indicating high phase purity. Scanning electron microscopy (SEM) showed that all four Al-MOFs had nanosheet-like crystal morphologies. Transmission electron microscopy (TEM) and elemental mapping of MOF-303 confirmed its crystal formation and uniform elemental distribution, verifying structural integrity. Thermogravimetric (TG) analysis showed that all Al-MOFs had high thermal stability, maintaining constant weight up to 653-693 K.
2. Porosity characterization:
N₂ adsorption-desorption isotherms at 77 K showed that the four Al-MOFs all exhibited Type I isotherm behavior, characteristic of microporous materials, with micropore sizes in the range of 4.1 Å to 5.2 Å. MOF-303 had the highest N₂ uptake at different relative pressures, with a Brunauer-Emmett-Teller (BET) specific surface area of 1335 m²·g⁻¹, which was consistent with reported literature and simulated results. After the first deionized water washing regeneration, the BET surface area of MOF-303 decreased to 1072 m²·g⁻¹, and recovered to 1184 m²·g⁻¹ after alkaline solution washing in the fifth cycle.
3. Spectroscopic characterization:
In situ diffuse reflectance infrared Fourier-transform spectroscopy (in situ DRIFTS) of MOF-303 during NO₂ adsorption showed a peak at 1730 cm⁻¹ (characteristic of NO₂ physisorption), a gradual decrease in the μ₂-OH band at 3700 cm⁻¹ and the emergence of an O-H stretching peak at 3650 cm⁻¹ (indicating hydrogen bonding), a negative peak at 1660 cm⁻¹ (CN bond depletion) and a peak at 925 cm⁻¹ (dipole interaction between NO₂ and pyrazole -N sites). Near-ambient pressure X-ray photoelectron spectroscopy (NAP-XPS) showed a ~0.5 eV positive shift in the N 1s peak of MOF-303 after NO₂ exposure and a new peak at 406.2 eV, confirming the presence of intact physisorbed NO₂ molecules and electron withdrawal due to dipole interactions.
4. Practical performance characterization:
Breakthrough experiments showed that MOF-303 had a NO₂ retention time of 821 min·g⁻¹ under dry conditions and 1107 min·g⁻¹ at 50% RH, with enhanced adsorption capacity in humid environments. Cyclic adsorption experiments showed that MOF-303 maintained 100%, 92%, 85% and 73% of its dynamic NO₂ capacity in the first four regeneration cycles, and recovered to 90% in the fifth cycle after alkaline washing. PXRD patterns remained essentially unchanged throughout the cycling tests, confirming the structural stability of MOF-303.
Mechanism Analysis
1. Electrostatic potential and interaction mechanism:
The polar NO₂ molecule has a strong dipole moment (two electronegative O atoms and one electropositive N atom), and its interaction with MOFs is determined by the electronic nature of heteroatom functional sites. MOF-303's -N-NH bifunctional sites realize synergistic effects: the N atom of NO₂ forms dipole-induced dipole interactions with the electronegative -N sites of PZDC linkers, and the O atoms of NO₂ form hydrogen bonds with the electropositive -NH sites of PZDC linkers and the μ₂-OH groups of AlO₆ octahedral chains. MIL-160 and CAU-23 only form moderate dipole interactions with NO₂ through -O and -S sites respectively, while KMF-1 only has weak hydrogen bonding through electropositive -NH sites, resulting in a gradual increase in NO₂ adsorption capacity in the order of -NH < -S < -O < -N-NH.
2. Theoretical calculation verification:
DFT calculations revealed two main NO₂ binding sites in MOF-303: at Site I, NO₂ is parallel to the PZDC linker, forming dipole interactions (N_NO₂···N_PZDC = 3.30 Å), hydrogen bonds (2.84 Å and 3.04 Å) and weak van der Waals interactions (N_NO₂···π_PZDC = 4.43 Å) with binding energy of -23.9 kJ·mol⁻¹; at Site II, NO₂ forms hydrogen bonds with μ₂-OH groups (O_NO₂···H_AlO₆-octahedra = 1.82 Å) and -NH sites (O_NO₂···H_PZDC = 2.53 Å), and dipole interactions (N_NO₂···N_PZDC = 4.19 Å) with binding energy of -41.3 kJ·mol⁻¹. Ab initio molecular dynamics (AIMD) simulations in the 0-5000 fs window confirmed that NO₂ had the most stable anchored interaction on MOF-303, while the other three Al-MOFs showed weaker and less stable NO₂ interactions.
3. NO disproportionation and adsorption species:
All four Al-MOFs exhibited low NO release during NO₂ adsorption, and the NO release level declined after the initial plateau, indicating that only a small fraction of NO₂ underwent chemical disproportionation (2NO₂ → NO + NO₃⁻), and subsequent NO₂ adsorption was dominated by physisorption. In situ DRIFTS did not detect peaks related to N₂O₄, indicating that the micropore nanoconfinement of MOF-303 restricted NO₂ dimerization, and monomeric NO₂ was the main adsorption species, which was stabilized by specific host-guest interactions.
Summary
1. The research team developed a NO₂-specific high-throughput computational screening strategy guided by physicochemical descriptors, which integrated pore accessibility, framework polarity, acid-resistant robustness and synthetic feasibility, and screened out four Al-MOFs with different heteroatom functional sites from more than 15,000 MOFs in the CoRE database, breaking the limitation of single geometric factor screening in previous studies.
2. The four synthesized Al-MOFs (KMF-1, CAU-23, MIL-160, MOF-303) have high phase purity, microporosity and thermal stability, and MOF-303 with -N-NH bifunctional sites exhibits an outstanding dynamic NO₂ adsorption capacity of 5.31 mmol·g⁻¹, which is significantly higher than that of reported benchmark porous adsorbents, and shows enhanced adsorption performance in humid environments.
3. Multiple characterization techniques (in situ DRIFTS, NAP-XPS) and theoretical calculations (DFT, AIMD) revealed that the superior NO₂ adsorption performance of MOF-303 is attributed to the synergistic dipole interaction and hydrogen bonding at the -N-NH bifunctional sites, and the micropore nanoconfinement effect stabilizes monomeric NO₂ and reduces NO disproportionation, realizing moderate and reversible NO₂ adsorption.
4. MOF-303 has excellent regenerability and structural stability: it maintains good adsorption capacity after multiple cyclic regeneration, and alkaline solution washing can effectively restore its adsorption performance and specific surface area; its synthesis uses low-cost commercial precursors and non-toxic solvents, with high yield and scalability, and environmental and economic evaluation shows good sustainability.
5. This study establishes a molecular-level closed-loop chemical design workflow from theoretical prediction (HTCS) to experimental validation and mechanism analysis, which not only verifies the structure-adsorption correlation of Al-MOFs for NO₂ capture, but also provides a transferable research paradigm for the discovery of functional MOFs for targeted toxic gas capture and environmental remediation.
High-Throughput Screening Assisted Discovery of Robust Metal–Organic Frameworks for Efficient Ambient NO2 Removal
Authors: Jianqing WuShanshan Shang*Xuquan LiuJinpeng ZhangChao YangFengliang WangFengying MaXinyu ChenGaozhou LiangHaitao ZhouXiangwei ZouPeirong ChenHongxia XiQinfen Gu*Chongxiong Duan*Daiqi Ye*
DOI: 10.1021/jacs.5c21490
Links: https://doi.org/10.1021/jacs.5c21490
The above review is for academic progress sharing. For any errors or copyright issues, please contact us for correction or removal.



