Home >
News > Room-Temperature Separation of Krypton and Xenon Using a Flexible Coordination Framework Adsorbent
Room-Temperature Separation of Krypton and Xenon Using a Flexible Coordination Framework Adsorbent
Xenon-krypton separation is critical for high-tech, national defense, aerospace and nuclear energy fields. Adsorptive separation with porous solids is a promising alternative to energy-intensive cryogenic processes, but traditional adsorbents preferentially adsorb xenon and require high desorption energy for pure xenon production. This study reports a metal-organic framework (MOF) with synergistic structural and local flexibility that reverses this selectivity, enabling preferential krypton adsorption at room temperature. The material (MIP-203-Suc) achieves a krypton uptake of 36.8 cm³·cm⁻³ (298 K, 1 bar) and a Kr/Xe selectivity of 10.4 in 1/99 Kr/Xe breakthrough experiments, exhibits excellent radioactive stability, and efficiently captures 40 ppm trace krypton. Mechanistic studies show that dynamic cavity window adjustment and local ligand vibration synergistically exploit the slight size difference between Kr and Xe, realizing kinetically controlled Kr sieving via transiently expanded channels. This energy-efficient strategy redefines noble gas purification material design and provides new ideas for separating dynamically matched molecular mixtures.
Research Background
1. Core Problems:
Xenon is a high-value noble gas with surging demand, mainly produced via energy-intensive cryogenic separation from air and nuclear exhaust, where Kr is a persistent impurity. Xe (4.047 Å) and Kr (3.655 Å) have similar sizes, making size sieving difficult; traditional adsorbents thermodynamically adsorb Xe (higher polarizability: 40.44 × 10⁻²⁵ cm³ vs. 24.84 × 10⁻²⁵ cm³ for Kr) but require high energy for Xe desorption. Nuclear exhaust contains 40 ppm radioactive ⁸⁵Kr (10.8-year half-life), and its efficient room-temperature capture is an unmet environmental need.
2. Existing Solutions:
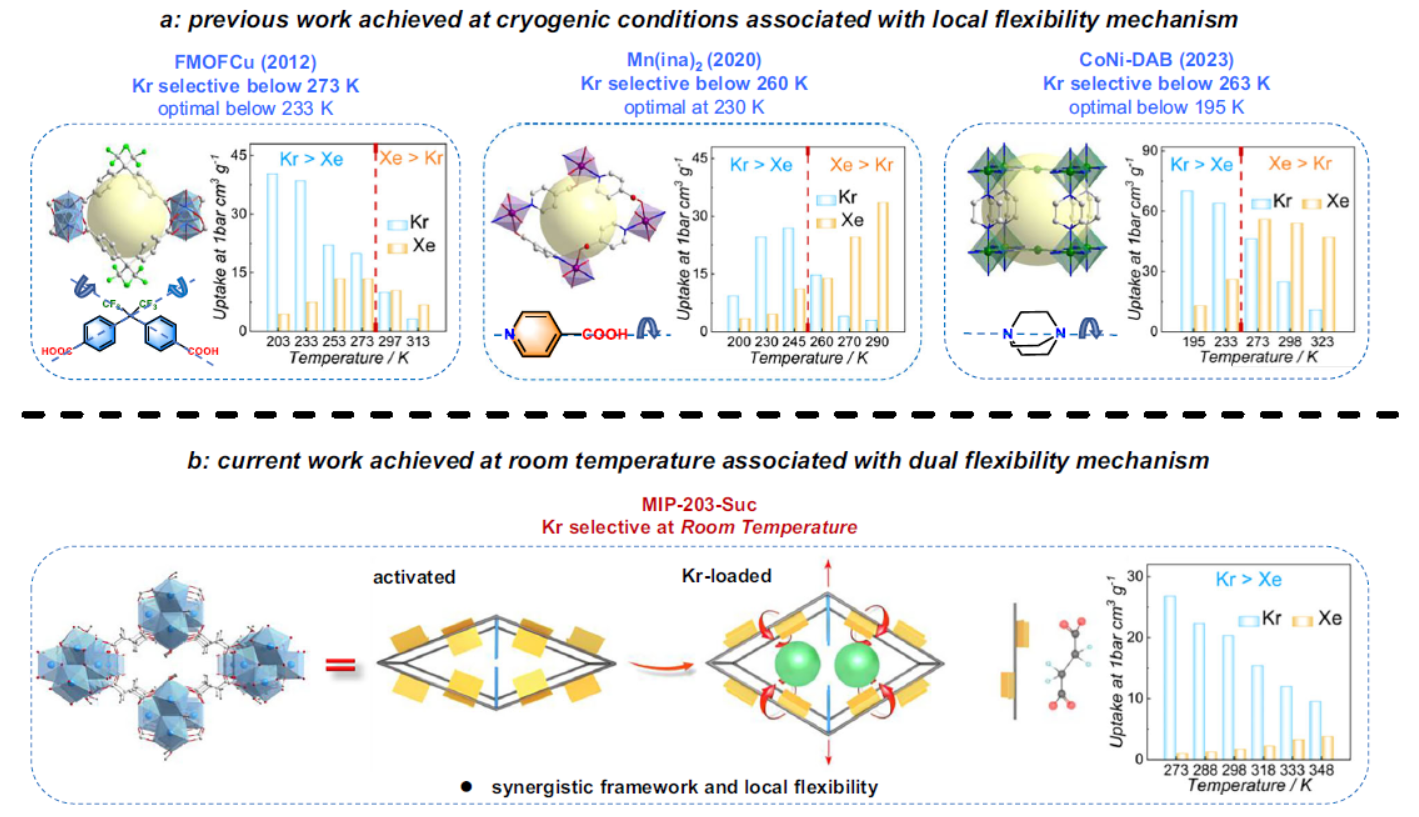
A small number of porous materials (FS-CuBTC_UM, CSuc-750 carbon, FMOFCu/Mn(ina)₂/CoNi-DAB MOFs) achieve Kr selectivity via kinetic sieving but only at temperatures below 273 K, switching to Xe selectivity at room temperature. Guest-responsive dynamic MOFs with narrow pore windows show potential for Kr/Xe discrimination due to differential diffusion energy barriers, laying a foundation for room-temperature Kr-selective adsorbent design.
3. Innovations:
The authors proposed a synergistic structural and local flexibility design for coordination frameworks, selecting MIP-203-Suc—an ultrasmall-pore Zr-MOF based on succinic acid with temperature/guest-responsive dynamics and local bond flexibility. Thermal activation modulates its pore size to the optimal Kr/Xe separation range, and succinate’s aliphatic chain flexibility fine-tunes gas accessibility, enabling the first room-temperature preferential Kr adsorption and kinetic sieving, while solving trace radioactive Kr capture and high-purity Xe preparation.
Experimental Content
1. Synthesis of MIP-203-Suc:
A mixture of ZrCl₄ (233 mg, 1 mmol), succinic acid (500 mg, 4.23 mmol) and formic acid (3.8 mL) was heated in a 25 mL Teflon-lined autoclave at 393 K for 72 h. The resulting white powder was washed with water and methanol-exchanged for purification. The method uses low-cost, readily available precursors and mild conditions, suitable for potential scale-up.
2. Gas Adsorption Measurement:
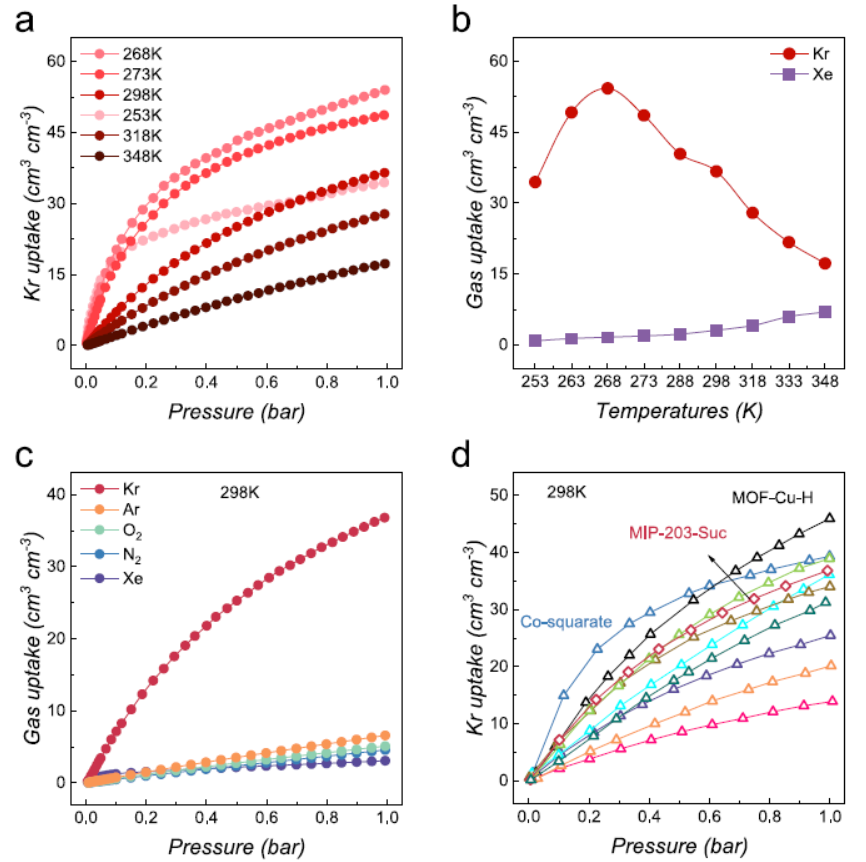
As-synthesized MIP-203-Suc (≥200 mg) was vacuum-activated at 393 K for 12 h. A Micromeritics 3Flex instrument measured single-component adsorption/desorption isotherms of ultra-high-purity gases (He, N₂, O₂, CO₂, Ar, Xe, Kr) at 273–348 K (0–100 kPa). Key Results: At 298 K/1 bar, Kr uptake reached 36.8 cm³·cm⁻³ (one of the highest reported for porous Kr adsorbents), far exceeding Xe-selective MOFs (Ag-MOF-303, HOF-FJU-46); Kr uptake was consistently higher than Xe at 253–348 K, with negligible adsorption of air components (Ar, O₂, N₂).
3. Column Breakthrough Experiments:
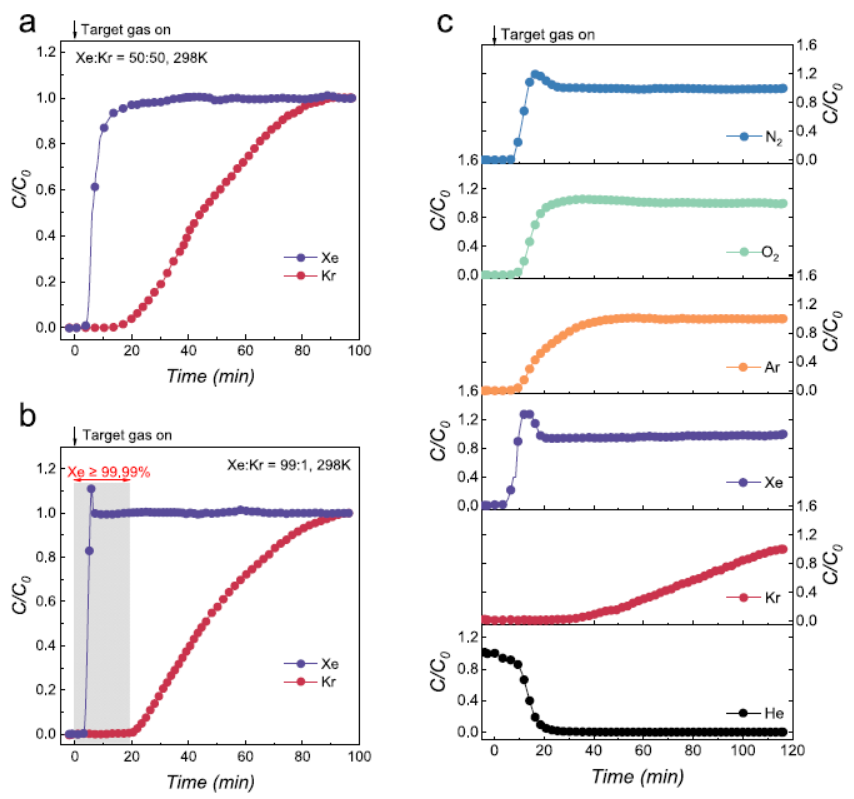
A copper column (5.0 mm ID, 300 mm L) packed with ~4.0 g activated MIP-203-Suc was vacuum-activated at 393 K for 12 h and He-presaturated. Effluent was monitored via mass spectrometry, and purity analyzed by gas chromatography. Results: (1) 50/50 Kr/Xe (298 K): Xe eluted at ~5 min, Kr retained for ~15 min, dynamic Kr capacity 0.40 mmol·g⁻¹, Kr/Xe selectivity 6.3; stable performance over 5 cycles, confirming regenerability. (2) 1/99 Kr/Xe: Xe eluted in <5 min, Kr retained for >22 min, Xe purity increased to 99.99% (100-fold improvement), selectivity rose to 10.4 (65% higher than equimolar mixture). (3) Simulated nuclear exhaust (400 ppm Xe + 40 ppm Kr + dry air): Kr trapped for 32 min, extraction rate 0.054 mmol·kg⁻¹; structural and separation performance intact after 100 kGy γ-irradiation.
4. DFT Calculations:
Climbing-image nudged elastic band (CI-NEB) calculations via VASP 6.4 (PBE-GGA, DFT-D3/BJ) studied Kr/Xe diffusion paths. Results: The most favorable a-axis Path-a had diffusion energy barriers of 12.8 kJ·mol⁻¹ (Kr) and 21.0 kJ·mol⁻¹ (Xe); at 298 K/50 kPa, Kr’s diffusion time constant (9.37 × 10⁻³ min⁻¹) was far higher than Xe’s (7.38 × 10⁻⁴ min⁻¹), with a kinetic selectivity of 12.7.
Characterization and Analysis
1. X-ray Diffraction (PXRD/Synchrotron PXRD):
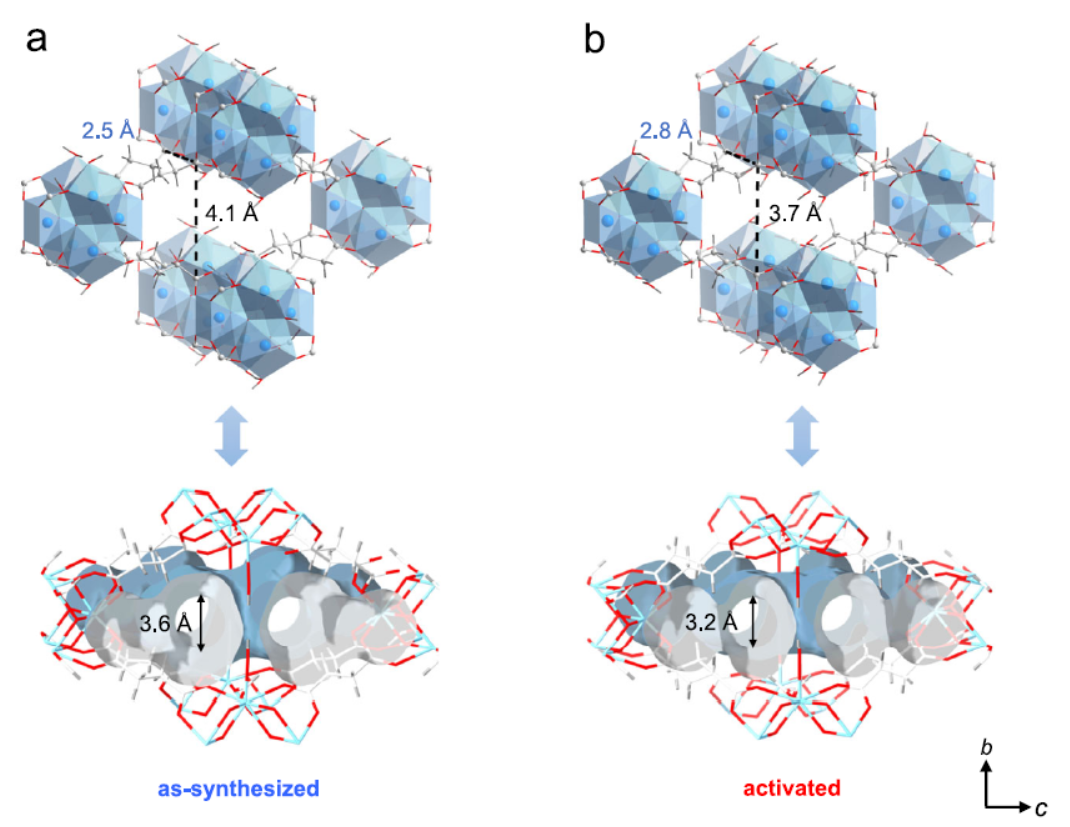
Routine PXRD (Bruker D8 Advance) and synchrotron PXRD (SSRF BL14B1, 0.6887 Å) characterized structural changes. Results: 373 K thermal activation caused anisotropic pore modulation: a-axis window shrank from 4.1 Å (as-synthesized) to 3.7 Å (activated), b-axis aperture expanded from 2.5 Å to 2.8 Å. Kr-loaded PXRD matched the as-synthesized phase, confirming reversible structural flexibility (in contrast to rigid CO₂ adsorption behavior). High-resolution refinement showed Kr atoms near succinate linkers (Kr-H: 3.52–3.70 Å), far from Zr₆ cluster hydroxyls, minimizing interatomic repulsion.
2. Pore Property Characterization:
CO₂ adsorption at 195 K determined permanent porosity, with Horvath-Kawazoe (H-K) pore size analysis. Results: Activated MIP-203-Suc had a BET surface area of 161 m²·g⁻¹ and bimodal pore size distribution (peaks at ~4.0 Å and ~5.0 Å), consistent with crystallographically resolved 1D bottle-shaped channels—the structural basis for Kr/Xe sieving.
3. Raman Spectroscopy:
A Horvath LabRAM HR Evolution spectrometer (633 nm laser) characterized Kr-induced structural changes. Results: Kr loading caused succinate carboxylate vibration peaks to redshift (1604→1614 cm⁻¹, 1468→1479 cm⁻¹) and a -CH₂/C-C-C peak to blueshift (1284→1276 cm⁻¹), confirming Kr-assisted local flexibility of succinate linkers, which mediates Kr migration.
4. Diffusion Kinetics:
The classical micropore diffusion model calculated kinetic parameters. Results: Xe’s diffusion time constant was highly temperature-dependent (3.44 × 10⁻⁴ min⁻¹ at 273 K → 8.77 × 10⁻⁴ min⁻¹ at 318 K), while Kr’s only slightly increased (1.72 × 10⁻³ → 2.03 × 10⁻³ min⁻¹), showing larger Xe molecules are more sensitive to thermal activation due to higher diffusion barriers—key to temperature-dependent kinetic separation.
Mechanism Analysis
The authors combined experimental characterization and DFT calculations to reveal the room-temperature kinetically controlled Kr sieving mechanism of MIP-203-Suc, centered on the synergism of structural and local ligand flexibility:
1. Thermally Induced Anisotropic Pore Modulation:
MIP-203-Suc is a Zr-MOF with 10-coordinated Zr₆ clusters and succinate linkers (flexible C4 backbone). Reduced Zr₆ connectivity (vs. 12-coordinated rigid analogues) enhances framework flexibility and ligand mobility. 373 K activation shrinks the a-axis window to 3.7 Å (matching Kr’s kinetic diameter) and slightly expands the b-axis, creating a pore structure that sterically hinders larger Xe.
2. Dual-Mode Flexibility and Transient Pore Expansion:
The material has framework flexibility and local succinate linker flexibility (rotation/stretching). Kr passage induces succinate torsional rotation: the O-C-O/C-C-C dihedral angle increases from 20.5° (activated) to 34.8° (Kr crossing), and aliphatic chain C-C distance expands from 5.6 Å to 6.66 Å, enabling transient pore expansion. This deformation is fully reversible, ensuring structural stability. Xe requires a 40.9° dihedral angle (sacrificing ligand geometry), leading to a much higher diffusion barrier and kinetic discrimination.
3. Anisotropic Diffusion and Energy Barrier Differences:
DFT/CI-NEB calculations identified 6 viable diffusion paths, with a-axis Path-a as the most favorable. The 8 kJ·mol⁻¹ energy barrier difference (Kr vs. Xe) causes Kr’s far faster diffusion, the core of kinetic sieving; b-axis paths have much higher barriers, so mass transfer is dominated by the a-axis.
4. Kr-Selective Framework Response:
Raman and PXRD results confirm MIP-203-Suc has a selective structural response to Kr: Kr adsorption activates succinate’s local flexibility and induces reversible framework distortion, while the material is rigid in CO₂ adsorption—host-guest interactions between the framework and Kr drive this flexibility, enhancing Kr selectivity.
Summary
1. The authors synthesized MIP-203-Suc, a flexible Zr-MOF with synergistic structural and local flexibility, via a simple solvothermal method using ZrCl₄ and succinic acid as precursors.
2. This material achieves the first room-temperature preferential Kr adsorption and kinetic sieving among MOFs, with a high Kr uptake (36.8 cm³·cm⁻³, 298 K/1 bar) and Kr/Xe selectivity up to 10.4 for trace Kr separation (1/99 Kr/Xe).
3. MIP-203-Suc efficiently captures 40 ppm trace Kr in simulated nuclear exhaust, increasing Xe purity from 99% to 99.99% in a single cycle; it has excellent radioactive stability (100 kGy γ-irradiation resistance), solving a key nuclear industry challenge.
4. The core mechanism of its excellent performance is the synergism of thermally induced anisotropic pore modulation, dual-mode flexibility-mediated transient pore expansion, and significant Kr/Xe diffusion energy barrier differences; reversible structural flexibility ensures reusability.
Krypton/Xenon separation at room temperature in a flexible coordinative framework sorbent
Authors: Wenchang Wang, Jiahao Lou, Qiang Wu, Biao Hu, Jun Wang, Xiangbiao Yin*, Chengliang Xiao*
DOI: 10.1038/s41467-025-67537-3
Links: https://www.nature.com/articles/s41467-025-67537-3
The above review is for academic progress sharing. For any errors or copyright issues, please contact us for correction or removal.



